Overview
Source: Laboratory of Jeff Salacup - University of Massachusetts Amherst
The product of an organic solvent extraction, a total lipid extract (TLE), is often a complex mixture of hundreds, if not thousands, of different compounds. The researcher is often only interested in a handful of compounds or, if interested in many, may need to remove unwanted constituents that are"in the way" or co-eluting. For example, the concentrations of individual compounds in a sample are often determined on a gas chromatograph coupled to a flame-ionizing detector (GC-FID), because the relationship between FID response (in pA) and the amount of compound in a sample (e.g., ng/µL) is both linear and sensitive. The GC portion of the instrument separates different compounds in a sample based on their boiling point, chemical structure, and affinity with a solid phase that can change according to application. The result is a chromatogram (Figure 1), showing the separation of different chemical constituents in time, as well as their relative concentration (calculated as the area under the curve). However, sometimes more than one compound elutes off the GC at a time (Figure 1). In this case, sample purification is required before compounds can be confidently quantified.
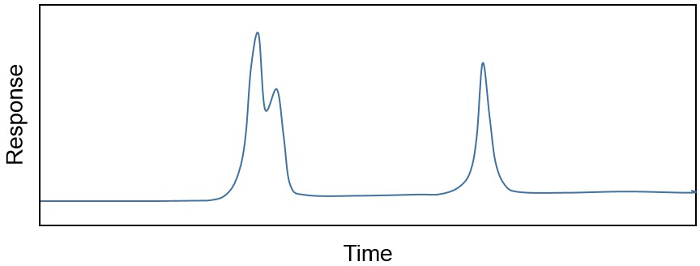
Figure 1. A chromatogram showing the separation of different chemical constituents over time and their relative concentration (area under the curve). Co-eluting and separated peaks are shown.
Principles
FAMEs (fatty acid methyl esters) are important constituents of terrestrial and marine microbes and are often used, in conjunction with genetic techniques, to describe ecosystem microbial diversity in modern and ancient systems. However, the presence of FAMEs in a sample is not always helpful. Due to their similar size and chemistry, FAMEs called alkenonatescommonly co-elute with long-chain alkyl ketones, called alkenones (Figure 2). The distribution of alkenones in a sample relates information on sea surface temperature at the time and/or the place the sample was taken, and so their accurate and precise characterization is important.
Saponification is a common purification technique used to convert FAMEs into fatty acids, thus changing their chemical characteristics enough to remove them from co-elution with alkenones (Figure 3). Saponificationis a form of hydrolysis. In hydrolysis, water is used to split molecules. Saponification is a hydrolysis that is accelerated in the presence of a base, such as potassium hydroxide (KOH). KOH dissolves into K+ and OH- in water. The hydroxide anion (OH-, negatively charged ion) adds to the slightly, positively charged tertiary carbon atom at the heart of the FAME (Figure 3, top). However, this chemical configuration is unstable, (carbon has bonded to too many other atoms) and the alkoxide (ROH-) is expelled. But the H of the conjugate base this expulsion forms quickly moves to the alkoxide to form the alcohol methanol and fatty acid potassium salt (Figure 3, middle). At this point, the offending FAME (alkenoate) has been converted to a chemical that no longer co-elutes with it. However, if one wishes to also analyze FAME chemistry, they can be reclaimed by the addition of acid (HCl) to the solution, until pH reaches ~2. At this pH, the fatty acid potassium salt is split to form a carboxylic acid and ionic salt (KCl; Figure 3, bottom).

Figure 2. The chemical structures of an alkenone with 37 carbon atoms and 2 double bonds (top) and its associated alkenoate FAME (bottom).

Figure 3. A schematic of the saponification of palmitic acid using potassium hydroxide (KOH) to increase the rate of hydrolysis (http://www.mpbio.com/).
Saponification is a technique commonly used to remove fatty acid methyl esters from a complex organic mixture.
Complex organic samples are often analyzed by gas chromatography, which is used to determine the relative concentrations of individual components.
However, compounds that are similar in size and structure cannot be distinguished by the instrument, skewing the results. Therefore, unwanted compounds that produce overlapping signals must be removed in order to obtain accurate results.
This video covers the use of saponification to purify alkenones for paleoclimatology. It is the first in a series detailing the purification of complex biomarker samples. It will cover the procedure, as well as some other uses of the technique.
Polyunsaturated long chain alkyl ketones, called alkenones, have been shown to provide useful information about past sea surface temperature.
However, the organisms that produce alkenones often create fatty acid methyl esters that are similar in size and chemical structure, called alkenoates. Because of these similarities, alkenoates must be removed before an accurate analysis can be obtained.
Saponification is a common technique used to prevent co-elution of these molecules. Saponification utilizes water to split the molecular bond of an ester. A base attaches to the carbon at the heart of the alkenoate. This addition reaction creates an unstable intermediate, and the alkoxide is expelled.
The hydrogen from the newly formed acid moves to the expelled alkoxide, and the resulting carboxylate anion forms an ionic bond with the cation from the base. The result is an alcohol and a fatty acid salt. Adding a strong acid will re-generate the carboxylic acid. At this point, the offending alkenoate has been converted to a form that no longer co-elutes with the alkenone of interest.
Now that you understand how saponification can be used to purify an organic mixture, you are ready to begin the procedure.
First, acquire a dried total lipid extract - or TLE - that was obtained using a solvent extraction method. Next, prepare the saponification solutions as outlined in the text protocol. Ensure that all components are pure and free of hydrocarbons. Once the solutions are prepared, add the dried TLE to a 60-mL borosilicate glass vial. Add 2 mL of 2 normal potassium hydroxide, and seal the vial. Next, heat the vial to 60 °C for 2.5 h to cleave the ester bond. When this is complete, allow the sample to cool to room temperature. Once the sample has cooled, add 2 mL of 5% sodium chloride solution to the vial. Cap the vial, and shake briefly. Add 6 N hydrochloric acid dropwise until a pH of 2 is reached - using pH paper to test. The addition of this acid will protonate the carboxylate anion to form the final product - the stable carboxylic acid.
Now that the acidified solution is ester-free, add 5 mL of hexane. Cap and shake vigorously for 5 seconds to extract the organic compounds from the water. Allow the solution to rest until the organic and aqueous phases separate completely. Salts, ions, and unreacted hydrochloric acid will remain in the aqueous phase, while organic compounds will separate into the hexane. Once the phases have completely separated, remove approximately 75% of the hexane using a pipette, and dispense into another 40-mL vial. Repeat this extraction process two additional times, adding 5 mL of hexane each time. Once this is complete, discard the leftover aqueous solution into a proper waste container. Label the vial containing the freshly saponified organic phase. The carboxylic acids produced cannot be injected into the instruments commonly used for alkenone analysis without further purification. Carboxylic acids injected into a gas chromatograph quickly accumulate and ruin inlets, inlet liners, and the front end of the columns. To remove these acids, the sample first needs to undergo another purification technique: separation via column chromatography.
Saponification has several applications in the extraction and purification of organic molecules.
Saponification can be used not only to separate biomarkers, but also to extract individual components for use in commercial products. In this example, compounds from the tobacco tree - Nicotiana glauca - were isolated and analyzed to investigate their potential as feedstock for a wide variety of bio-based products, such as fuel, heat, and an array of chemical compounds.
The leaves were first homogenized, then centrifuged, to concentrate the molecules of interest. The concentrated plant material was then saponified. The extracted material was analyzed with liquid chromatography-mass spectrometry, to determine the concentration of tocopherol - a family of vitamin E compounds typically found in plants.
The extracted material can also be reconstituted in vitro to its original composition. In this example, saponification was used to extract carotenoids from spinach plants, to be later reconstituted in vitro. The spinach was first homogenized, then centrifuged, to harvest the pigment molecules. These molecules were then suspended in a solution of potassium hydroxide in a separatory funnel, initiating saponification. The saponified carotenoids separated into an ether layer, which was collected and dried. Using a series of solution buffers, the carotenoids - and other pigmentation molecules - were later reconstituted in vitro. This purification allowed for analysis of these pigments without the interference of similarly structured organic compounds.
Because of its ability to hydrolyze esters, saponification can be used to "free" compounds that are otherwise bound to macromolecules. In this example, an anhydrous saponification step is used to convert ethyl 4-fluorobenzoate to the carboxylic acid salt of potassium4-fluorobenzoate. This deprotection through saponification allows for the production of crude SFB - a reaction that wouldn't be possible had the molecule remained "stuck".
You've just watched JoVE's introduction to the purification of Uk'37 samples through saponification. You should now understand how saponification works, and how to use it to purify alkenones in a total lipid extract. Further purification processes will be demonstrated in subsequent videos.
Thanks for watching!
Subscription Required. Please recommend JoVE to your librarian.
Procedure
1. Setup and Preparation of Materials
- Obtain a total lipid extract (TLE) using a solvent extraction method (Sonication, Soxhlet, or Accelerated Solvent Extraction (ASE)).
- Prepare a solution of 2 N KOH in 5% H2O in methanol.
- KOH and methanol can be purchased from chemical retailers. These chemicals should be pure and free of hydrocarbons.
- The molar mass of KOH is ~56 amu yielding 1 mol of OH- for each mol of KOH. Thus, to achieve a 2 N concentration, dissolve 112 g of KOH in 0.95 L of pure water and 0.05 L of methanol.
- Note that the dissolution of KOH in water is exothermic and creates a large amount of heat. Take care to add the KOH pellets to the water slowly to avoid a violent reaction.
- Dissolve 112 g of KOH pellets into 0.95 L of pure water on an automatic stir plate.
- Add 0.05 L of methanol once all of the KOH is dissolved to aid in the dissolution of the organic biomarkers into the aqueous solution.
- KOH and methanol can be purchased from chemical retailers. These chemicals should be pure and free of hydrocarbons.
- Prepare a solution of 6 N hydrochloric acid (HCl) in H2O.
- HCl can be purchased from chemical retailers. This chemical should be pure and free of hydrocarbons.
- HCl generally comes concentrated as 13 N. Thus, a 1:1 mixture of HCl and pure water produces a 6.5 N mixture of HCl, which is close enough to 6 N for the purposes of this experiment.
- Be sure to add the HCl to the water and not the other way around, as adding water to concentrated HCl is exothermic and generates heat. This may cause HCl to splash.
- Gently and slowly pour 100 mL HCl into a beaker containing 100 mL pure water,swirling the mixture between additions of HCl.
- Prepare a solution of 5% NaCl (table salt) in H2O.
- NaCl can be purchased from chemical retailers. This chemical should be pure and free of hydrocarbons.
- Calculate and weigh the mass of salt needed to make ~1 L of 5% solution (w/w). 1 L of water weighs ~1 kg or 1,000 g. Thus, 50 g of NaCl dissolved into 950 mL gives a 5% solution (50 g+ 950 g = 1,000 g; 50 g / 1,000 g = 0.05 or 5%).
- Gently add 50 g of NaCl to pure water on an automatic stir plate and wait for it to dissolve.
- Obtain the following materials: 2 clean and combusted (550 ˚C for 6 h) 40 mL borosilicate glass vials with PTFE-lined cap; a warming oven or heating blocks; combusted (550 ˚C for 6 h) borosilicate glass pipettes and bulbs; pH tape (acidic range); Hexane (hexane can be purchased from chemical retailers. This chemical should be Optima Grade or equivalent).
2. Methods
- Start with the dried TLE (containing esters) in one of the 40 mL borosilicate glass vials.
- Add approximately 10 mL of 2 N KOH to the TLE and cap.
- Heat in the oven or on a heating block to 60 ˚C for 2.5 h to cleave the ester bond (Figure 3).
- Remove the sample from heat and allow it to cool to room temperature.
- Add approximately 10 mL of the 5% NaCl solution to the TLE (Figure 3). This helps keep the interface between the aqueous and organic phase (to be added) from foaming. Cap and shake briefly.
- Add 6 N HCl to the salted TLE dropwise until pH 2 is reached to protonate the O- and form the final product, a stable carboxylic acid (Figure 3; use pH paper to test). If the TLE was colored,there may be a shift in color coinciding with pH 2.
- Add approximately 20 mL of hexane to the acidified solution, which is now ester-free. Cap and shake vigorously for 5 s to extract organic compounds from the water.
- Allow to set until aqueous and organic phases completely separate. Salts, ions, water, and unreacted HCl remain in the aqueous phase. Organic compounds are now in the hexane.
- Remove approximately 75% of the supernatant hexane using a pipette and dispense into the other 40 mL borosilicate glass vial.
- Repeat 2.7 - 2.9 twice, adding approximately 10 mL of hexane each time.
- Discard leftover aqueous mixture in a proper waste container.
- Label the vial containing hexane and ester-free organics "TLE - saponified.
Saponification is a technique commonly used to remove fatty acid methyl esters from a complex organic mixture.
Complex organic samples are often analyzed by gas chromatography, which is used to determine the relative concentrations of individual components.
However, compounds that are similar in size and structure cannot be distinguished by the instrument, skewing the results. Therefore, unwanted compounds that produce overlapping signals must be removed in order to obtain accurate results.
This video covers the use of saponification to purify alkenones for paleoclimatology. It is the first in a series detailing the purification of complex biomarker samples. It will cover the procedure, as well as some other uses of the technique.
Polyunsaturated long chain alkyl ketones, called alkenones, have been shown to provide useful information about past sea surface temperature.
However, the organisms that produce alkenones often create fatty acid methyl esters that are similar in size and chemical structure, called alkenoates. Because of these similarities, alkenoates must be removed before an accurate analysis can be obtained.
Saponification is a common technique used to prevent co-elution of these molecules. Saponification utilizes water to split the molecular bond of an ester. A base attaches to the carbon at the heart of the alkenoate. This addition reaction creates an unstable intermediate, and the alkoxide is expelled.
The hydrogen from the newly formed acid moves to the expelled alkoxide, and the resulting carboxylate anion forms an ionic bond with the cation from the base. The result is an alcohol and a fatty acid salt. Adding a strong acid will re-generate the carboxylic acid. At this point, the offending alkenoate has been converted to a form that no longer co-elutes with the alkenone of interest.
Now that you understand how saponification can be used to purify an organic mixture, you are ready to begin the procedure.
First, acquire a dried total lipid extract - or TLE - that was obtained using a solvent extraction method. Next, prepare the saponification solutions as outlined in the text protocol. Ensure that all components are pure and free of hydrocarbons. Once the solutions are prepared, add the dried TLE to a 60-mL borosilicate glass vial. Add 2 mL of 2 normal potassium hydroxide, and seal the vial. Next, heat the vial to 60 °C for 2.5 h to cleave the ester bond. When this is complete, allow the sample to cool to room temperature. Once the sample has cooled, add 2 mL of 5% sodium chloride solution to the vial. Cap the vial, and shake briefly. Add 6 N hydrochloric acid dropwise until a pH of 2 is reached - using pH paper to test. The addition of this acid will protonate the carboxylate anion to form the final product - the stable carboxylic acid.
Now that the acidified solution is ester-free, add 5 mL of hexane. Cap and shake vigorously for 5 seconds to extract the organic compounds from the water. Allow the solution to rest until the organic and aqueous phases separate completely. Salts, ions, and unreacted hydrochloric acid will remain in the aqueous phase, while organic compounds will separate into the hexane. Once the phases have completely separated, remove approximately 75% of the hexane using a pipette, and dispense into another 40-mL vial. Repeat this extraction process two additional times, adding 5 mL of hexane each time. Once this is complete, discard the leftover aqueous solution into a proper waste container. Label the vial containing the freshly saponified organic phase. The carboxylic acids produced cannot be injected into the instruments commonly used for alkenone analysis without further purification. Carboxylic acids injected into a gas chromatograph quickly accumulate and ruin inlets, inlet liners, and the front end of the columns. To remove these acids, the sample first needs to undergo another purification technique: separation via column chromatography.
Saponification has several applications in the extraction and purification of organic molecules.
Saponification can be used not only to separate biomarkers, but also to extract individual components for use in commercial products. In this example, compounds from the tobacco tree - Nicotiana glauca - were isolated and analyzed to investigate their potential as feedstock for a wide variety of bio-based products, such as fuel, heat, and an array of chemical compounds.
The leaves were first homogenized, then centrifuged, to concentrate the molecules of interest. The concentrated plant material was then saponified. The extracted material was analyzed with liquid chromatography-mass spectrometry, to determine the concentration of tocopherol - a family of vitamin E compounds typically found in plants.
The extracted material can also be reconstituted in vitro to its original composition. In this example, saponification was used to extract carotenoids from spinach plants, to be later reconstituted in vitro. The spinach was first homogenized, then centrifuged, to harvest the pigment molecules. These molecules were then suspended in a solution of potassium hydroxide in a separatory funnel, initiating saponification. The saponified carotenoids separated into an ether layer, which was collected and dried. Using a series of solution buffers, the carotenoids - and other pigmentation molecules - were later reconstituted in vitro. This purification allowed for analysis of these pigments without the interference of similarly structured organic compounds.
Because of its ability to hydrolyze esters, saponification can be used to "free" compounds that are otherwise bound to macromolecules. In this example, an anhydrous saponification step is used to convert ethyl 4-fluorobenzoate to the carboxylic acid salt of potassium4-fluorobenzoate. This deprotection through saponification allows for the production of crude SFB - a reaction that wouldn't be possible had the molecule remained "stuck".
You've just watched JoVE's introduction to the purification of Uk'37 samples through saponification. You should now understand how saponification works, and how to use it to purify alkenones in a total lipid extract. Further purification processes will be demonstrated in subsequent videos.
Thanks for watching!
Subscription Required. Please recommend JoVE to your librarian.
Results
This purification produces a TLE free of esters that may be co-eluting with the alkenones. However, the purification produced carboxylic acids, which cannot be injected onto instruments commonly used to analyze samples for alkenone concentrations because of their low volatility. For example, the boiling point of hexane, a 6-carbon hydrocarbon, is 68 ˚C, but the boiling point of its acid (hexanoic acid) is 205 ˚C. Most GC amenable biomarkers have from 20 to 35 carbon atoms (boiling point generally increases with an increasing number of atoms), and most GC temperature programs stop around 300 ˚C. Carboxylic acids injected into a GC quickly accumulate and ruin inlets, inlet liners, and the front end of columns. To remove these acids, the sample first needs to undergo another purification technique separation via column chromatography.
Subscription Required. Please recommend JoVE to your librarian.
Applications and Summary
As mentioned previously, saponification is commonly used in organic geochemistry labs to remove fatty acid methyl esters (FAMEs) of alkenones, called alkenoates, which co-elute with alkenones on gas chromatographs (Figure 1). Saponification is also used to "free" fatty acids "bound" to sediment or macromolecules.The degradation and preservation of organic matter and biomarkers in sediments involves the removal of functional groups (N, O, and S) and the eventual polymerization of individual biomarkers into macromolecules and/or the adsorption of biomarkers and macromolecules onto mineral surfaces. Not all biomarkers in a setting become bound, and the ratio of bound-to-free biomarkers may change by setting and sediment age for reasons still not fully explained. Saponification, sometimes at temperatures higher than those discussed here (>200 ˚C), is used to free these bound biomarkers in the effort to describe them, their source, and the mechanisms responsible for their bound nature.
Subscription Required. Please recommend JoVE to your librarian.




