11.18: 金属固体
铜,铝和铁晶体等金属固体由金属原子形成。 金属晶体的结构通常被描述为原子核在离域电子“海”中的均匀分布。 这种金属固体中的原子由一个独特的力 (称为金属键) 固定在一起,该体性质可产生许多有用且变化多样的。
所有金属固体都具有高热电导率和电导率,金属光泽度和可锻性。 许多人都是非常坚硬的的人,而且相当强大。 由于它们的可锻性 (在压力或锤击下变形的能力) ,它们不是碎落,因此它们制造了有用的建筑材料。 金属的熔点差别很大。 汞是室温下的液体,碱金属溶解在 200 °C 以下 几种过渡后金属的熔点含量也很低,而过渡金属在 1000 °C 以上的温度下熔化 这些差异反映了金属键的强度在金属之间的差异。
金属固体的性质
由于其结晶结构,金属固体几乎没有与结构相关的独特性质,并在下表中列出。
| 实体类型 | 粒子类型 | 吸引力类型 | 性质 | 示例 |
| 金属 | 电正元素原子 | 金属键 | 光亮、可锻、延性强、可良好地导热和电、可变硬度和熔化温度 | 铜、铁、铁、铅,U |
金属固体的晶体结构:密堆积
由相同原子组成的固体可以有两种类型的排列:正方形或密堆积 (图 1)。 由于密堆积可以最大限度地提高原子之间的整体吸引力,并将分子间总能量降至最低,因此大多数金属中的原子会以这种方式进行包装。

图 1. 立方对比密堆积排列。
我们在简单的金属晶体结构中发现两种最接近的堆积: 六角最密堆积(HCP) 和 立方最密堆积(CCP)。 这两个图层都由六边排列原子的重复图层组成。 在这两种类型中,第二层 (B) 放置在第一层 (A) 上,以便第二层中的每一个原子都与第一层中的三个原子接触。 第三层以两种方式之一定位。
在 HCP 中,第三层的原子直接位于第一层的原子之上 (即第三层也是 A 型) ,堆叠由交替的 A 型和 B 型密堆积层 (即 ABAB⋯) 组成 (图 2a)。
在 CCP 中,第三层中的原子不在前两层中的任何一层中的原子之上 (即第三层为 C 型) ,堆叠由交替的 A 型, B 型和 C 型密堆积层 (即 ABCABCBC⋯) 组成 (图 2b)。 立方面心(FCC) 和 CCP 排列实际上是相同的结构,具有紧凑型原子堆积,占体积的 74%。
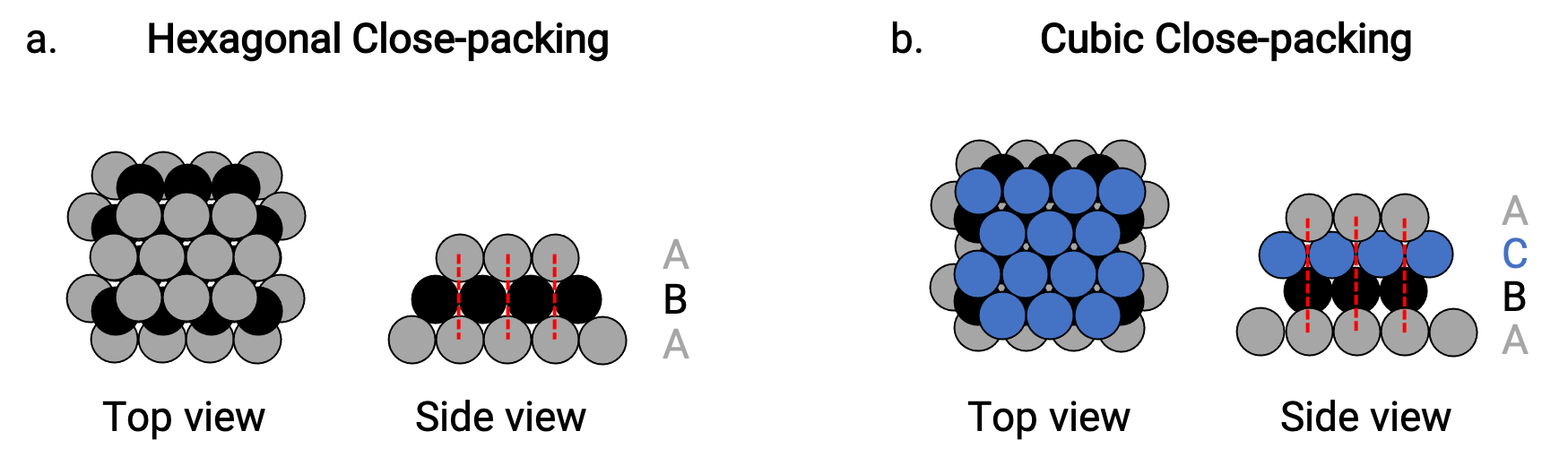
图 2. (a) 六角紧密堆积由两个交替层 (ABAB… ć ) 组成。 (b) 立方密堆积由三个交替层 (ABCABCABC…) 组成。
在这两 种类型的堆积中,每个原子接触其各自层中的六个原子,上面层中的三个原子,下面层中的三个原子。 因此,每一个原子接触 12 近邻,因此配位数为 12。
大约三分之二的金属在紧密堆积的阵列中结晶,配位数为 12。 HCP 结构中结晶的金属包括 CD , Co , Li , mg , Na , 和锌,以及在 CCP 结构中结晶的金属包括 Ag , Al , Ca , Cu , Ni , PB 和 Pt.
本文改编自 Openstax, 化学 2e, 第 10.5 节:物质的固态和 第10.6节: 结晶固体中的晶格结构。


