Summary
Total Vaporization Solid Phase Microextraction (TV-SPME) completely vaporizes a liquid sample whilst analytes are sorbed onto a SPME fiber. This allows for partitioning of the analyte between only the solvent vapor and the SPME fiber coating.
Abstract
Gas Chromatography – Mass Spectrometry (GC-MS) is a frequently used technique for the analysis of numerous analytes of forensic interest, including controlled substances, ignitable liquids, and explosives. GC-MS can be coupled with Solid-Phase Microextraction (SPME), in which a fiber with a sorptive coating is placed into the headspace above a sample or immersed in a liquid sample. Analytes are sorbed onto the fiber which is then placed inside the heated GC inlet for desorption. Total Vaporization Solid-Phase Microextraction (TV-SPME) utilizes the same technique as immersion SPME but immerses the fiber into a completely vaporized sample extract. This complete vaporization results in a partition between only the vapor phase and the SPME fiber without interference from a liquid phase or any insoluble materials. Depending upon the boiling point of the solvent used, TV-SPME allows for large sample volumes (e.g., up to hundreds of microliters). On-fiber derivatization may also be performed using TV-SPME. TV-SPME has been used to analyze drugs and their metabolites in hair, urine, and saliva. This simple technique has also been applied to street drugs, lipids, fuel samples, post-blast explosive residues, and pollutants in water. This paper highlights the use of TV-SPME to identify illegal adulterants in very small samples (microliter quantities) of alcoholic beverages. Both gamma-hydroxybutyrate (GHB) and gamma-butyrolactone (GBL) were identified at levels that would be found in spiked drinks. Derivatization by a trimethylsilyl agent allowed for conversion of the aqueous matrix and GHB into their TMS derivatives. Overall, TV-SPME is quick, easy, and requires no sample preparation aside from placing the sample into a headspace vial.
Introduction
Solid-Phase Microextraction (SPME) is a sampling technique in which a liquid or solid sample is placed into a headspace vial and a SPME fiber, coated with a polymeric material, is then introduced into the sample headspace (or immersed in a liquid sample). The analyte is sorbed onto the fiber and then the fiber is placed inside the GC inlet for desorption1,2. Total Vaporization Solid-Phase Microextraction (TV-SPME) is a similar technique as immersion SPME but completely vaporizes a liquid sample before analytes are adsorbed onto the fiber. This allows for partitioning of the analyte between only the solvent vapor and the coating of the fiber, allowing for more of the analyte to be adsorbed onto the fiber and resulting in good sensitivity3. There are various SPME fibers available and the fiber should be chosen based on the analyte of interest, solvent/matrix, and derivatization agent. See Table 1 for established TV-SPME analytes.
| Sample | Analyte(s) | Recommended SPME Fiber | Reference(s) |
| Human Hair | Nicotine, cotinine | Polydimethylsiloxane/divinylbenzene (PDMS/DVB), polyacrylate (PA) | 3 |
| Smokeless Powder | Nitroglycerin, diphenylamine | Polydimethylsiloxane (PDMS), polyethylene glycol (PEG) | 7, 8 |
| Racing fuel | Methanol, nitromethane | PEG | 9 |
| Water | Polycyclic aromatic hydrocarbons | PDMS | 10 |
| Beverages | ɣ-Hydroxybutyric acid, ɣ-butyrolactone | PDMS | This Work |
| Solid Powder | Methamphetamine, amphetamine | PDMS/DVB | Unpublished |
Table 1. Recommended SPME fibers with established TV-SPME analytes.
To perform TV-SPME, analytes are dissolved in a solvent and an aliquot of this mixture is placed into a headspace vial. Samples do not need to be filtered because only the solvent and volatile analytes will vaporize. Specific volumes of liquid samples must be used to ensure total vaporization of the sample. These volumes are determined by using the Ideal Gas Law to calculate the number of moles of a solvent multiplied by the molar volume of the liquid (Equation 1).
 Equation 1
Equation 1
where Vo is the volume of the sample (mL), P is the vapor pressure of the solvent (bar), Vv is the volume of the vial (L), R is the ideal gas constant (0.083145  ), M is the molar mass of the solvent (g/mol), T is temperature (K), and
), M is the molar mass of the solvent (g/mol), T is temperature (K), and  is the density of the solvent (g/mL).3
is the density of the solvent (g/mL).3
In order to use the correct vapor pressure, the Antoine equation (Equation 2) is used to account for the influence of temperature:4
 Equation 2
Equation 2
where T is temperature and A, B, and C are the Antoine constants for the solvent. Equation 2 may be substituted into Equation 1, yielding:
 Equation 3
Equation 3
Equation 3 gives the volume of the sample (Vo) that can be completely vaporized as a function of the temperature and solvent used.
To perform derivatization with TV-SPME, the SPME fiber is first exposed to a vial containing the derivatization agent for a predetermined amount of time depending on the analyte. The SPME fiber is then exposed to a new vial containing the analyte of interest. This vial is heated inside of a heated agitator. The analyte is then adsorbed onto the fiber with the derivatization agent. The derivatization of the analyte and/or the matrix takes place on the fiber before being inserted into the GC inlet for desorption. Figure 1 shows a depiction of the TV-SPME process with derivatization.

Figure 1: Depiction of the TV-SPME process with derivatization. The SPME fiber first enters the derivatization vial where the derivatization agent (yellow circles) sorb onto the fiber. The fiber is then introduced to the sample (blue circles) and heated. Formation of the derivative (green circles) takes place on the fiber during the extraction time. Please click here to view a larger version of this figure.
TV-SPME is beneficial because it allows for the analyte to be derivatized during the extraction process which reduces analysis time. Other methods, such as liquid injection, require that the analyte react with the derivatizing agent in solution prior to being injected into the GC. TV-SPME also requires little to no sample preparation. A matrix containing an analyte may be placed directly into the headspace vial and analyzed. Many compounds of interest are compatible with TV-SPME. Compounds must be soluble in a solvent and sufficiently volatile to allow for vaporization. Additionally, compounds must be thermally stable to be analyzed by GC-MS. TV-SPME has been used to analyze drugs and drug metabolites, racing fuels, polycyclic aromatic hydrocarbons, and explosive materials3,5,6,7,8,9,10.
Subscription Required. Please recommend JoVE to your librarian.
Protocol
1. General TV-SPME sample preparation and GC-MS analysis
NOTE: If the sample is already dissolved in a matrix, skip to Step 1.2.
- Extract or dissolve the solid sample in enough solvent (water, methanol, acetone, etc.) to reach the desired concentration. Liquid samples can be used “as is”.
NOTE: The amount of solid sample used depends on the desired concentration of the sample. Concentrations below 1 ppm (w/v) are recommended to avoid overloading the GC column. Analyte should be soluble in chosen organic solvent.- Ensure that the sample has fully dissolved.
- Calculate the volume needed to fully vaporize the sample using Equation 3 at the chosen temperature. For example, if the experiment is to be performed at 60 °C, calculate the volume needed to completely vaporize the solvent at 60 °C.
- Transfer this sample volume into a headspace vial and secure the cap. Acceptable methods for transferring liquid samples at the microliter scale include manually via glass syringe, an electronic glass syringe, or an autosampler robot capable of liquid transfers for sample preparation.
- If derivatizing the sample, prepare the proper derivatization agent by placing ~1 mL of the agent into a headspace vial.
- Choose the derivatization agent based on the type of derivatization needed: alkylation, acylation, or silyation. In this case, the recommended derivatization agent for the carboxylic acid and alcohol functional groups found on GHB is O-bis(trimethylsilyl) trifluoroacetamide (BSTFA). The derivatization agent may be used “as is” and does not require dilution. One mL of derivatization agent is enough to ensure complete saturation of the SPME fiber.
CAUTION: Derivatization agents are toxic and should be handled in a fume hood.
- Choose the derivatization agent based on the type of derivatization needed: alkylation, acylation, or silyation. In this case, the recommended derivatization agent for the carboxylic acid and alcohol functional groups found on GHB is O-bis(trimethylsilyl) trifluoroacetamide (BSTFA). The derivatization agent may be used “as is” and does not require dilution. One mL of derivatization agent is enough to ensure complete saturation of the SPME fiber.
- Set the proper incubation/extraction temperature based upon the calculation in step 1.2. This temperature ensures total vaporization, sufficient sample extraction, and complete derivatization (if necessary).
- Select GC-MS parameters (oven temperature program, flow rate, inlet temperature, etc.) based on the class of the compound(s) of interest. See Step 3 for an example parameter set.
- Ensure that the proper inlet liner (e.g., 2 mm inner diameter or less) is in the GC inlet.
- Ensure the SPME fiber has been properly conditioned and is in good working condition before beginning the analysis.
- Vary the conditioning parameters based on the type of SPME fiber being used. Please see SPME fiber instructions for proper conditioning temperature and time. In general, analyzing several SPME fiber blanks until they are reproducible is sufficient to characterize a SPME fiber as fully conditioned.
2. Gamma-hydroxybutyrate (GHB) and Gamma-butyrolactone (GBL) sample preparation
- Prepare a sample of GHB and/or GBL in water with a concentration of less than 1 ppm.
- Transfer 1 µL of this sample to a 20 mL headspace vial using one of the methods described in 1.2.1.
- Note that that the analysis of aqueous samples requires the lowest sample volumes. For example, one µL of water will completely vaporize into a 20 mL headspace vial at 60 °C.
- Cap the vial immediately.
- Place ~1 mL of BSTFA + 1% trimethylchlorosilane (TMCS) into a separate 20 mL headspace vial and cap.
NOTE: GBL does not derivatize. A derivatization step is still required, however, is to ensure the water solvent derivatizes and does not interfere with the sample.
CAUTION: BSTFA is toxic and should be handled in a fume hood.
3. GC-MS parameters and setup for GHB and GBL in water
- Create a method using the following GC-MS parameters:
Initial oven temperature: 60 °C held for 1 minute.
Oven program: 15 °C/minute.
Final oven temperature: 280 °C, held for 1 minute.
Flow rate: 2.5 mL/minute (speed optimized flow for a 0.25 mm i.d. column).
Inlet temperature: 250 °C.
Transfer line temperature: 280 °C. - Ensure a narrow (2 mm i.d. or less) SPME inlet liner has been placed inside the GC inlet.
- Ensure the PDMS/DVB SPME fiber has been properly conditioned and is in good working order prior to analysis.
NOTE: PDMS/DVB SPME fibers should be conditioned in the GC inlet at 250 °C for 30 minutes. PDMS/DVB SPME fibers should be an off-white color. - Run the GC-MS on the sample.
Subscription Required. Please recommend JoVE to your librarian.
Representative Results
A GBL volume study was performed to demonstrate the sensitivity of TV-SPME compared to headspace and immersion SPME. A 100ppmv sample of GBL in water was prepared and placed into 20 mL headspace vials with volumes of 1, 3, 10, 30, 100, 300, 1000, 3000, and 10,000 µL. The phase ratio of the samples allowed for TV-SPME (1-3 µL), Headspace SPME (10 – 3,000 µL) and Immersion SPME (10,000 µL). All samples were analyzed in triplicate and the average peak area was plotted against the sample volume. Overall, sample volumes that allowed for TV-SPME demonstrated more sensitivity than headspace or immersion SPME for GBL in water as shown in Figure 2. A comparison of the chromatograms for each method is shown in Figure 3.

Figure 2: Graph of average peak area versus sample volume for GBL in water. A GBL volume study was performed to demonstrate the efficacy of TV-SPME compared to headspace and immersion SPME. A 100ppmv sample of GBL in water was prepared and placed into 20 mL headspace vials with volumes of 1, 3, 10, 30, 100, 300, 1000, 3000, & 10000 µL. All samples were analyzed in triplicate and error bars correspond to standard deviation of the mean. Please click here to view a larger version of this figure.

Figure 3: Total Ion Chromatograms for GBL in water. (100 ppm) for 3 µL (blue), 300 µL (red), and 10,000 µL (green). Please click here to view a larger version of this figure.
Realistic samples of wine spiked with an effective dose of GHB and GBL are shown in Figure 4 and Figure 5, respectively. These samples also show the interconversion of GBL and GHB. When TV-SPME is performed properly, a sharp, abundant peak will result as shown in Figure 6. TV-SPME has good sensitivity and therefore proper concentrations should be used as to not overload the column. When high concentrations are present, peak asymmetry will result as shown in Figure 5 and Figure 7. In these cases, diluting the sample or using a split injection can improve peak shape.

Figure 4: Realistic sample of GHB in wine with an 8 mg/mL concentration. Peaks: 1) GBL, 2) hexanoic acid-TMS, 3) GHB-TMS2, 4) benzoic acid-TMS, 5) octanoic acid-TMS, 6) glycerol-TMS3, * denotes a cyclic siloxane (fiber/column bleed). Please click here to view a larger version of this figure.

Figure 5: Realistic sample of GBL in wine with a 10 mg/mL concentration. Peaks: 1) GBL, 2) hexanoic acid-TMS, 3) Siloxane, 4) trimethyl(2-phenylethoxy) silane, 5) GHB-TMS2. TIC shows GBL converting to GHB. Please click here to view a larger version of this figure.

Figure 6: Total Ion Chromatogram for GBL in water with a 0.1-ppm concentration. Results following the TV-SPME method previously described for GBL in water. Please click here to view a larger version of this figure.

Figure 7: Total Ion Chromatogram for GBL in water with a 10-ppm concentration. Results following the TV-SPME method previously described for GBL in water. Please click here to view a larger version of this figure.
When derivatizing, the analyst should ensure that the method allows for the analyte(s) to be fully derivatized prior to being desorbed into the GC. Partial derivatization can result in a peak representing the derivatized analyte and a peak representing the underivatized analyte. Partial derivatization will also result in a lower sensitivity for the analyte as less of it may adsorb onto the fiber.
Subscription Required. Please recommend JoVE to your librarian.
Discussion
TV-SPME has some benefits over liquid injection GC in that large sample sizes (e.g., 100 µL) may be used without instrument modifications. TV-SPME also has some of the same benefits as headspace SPME. Headspace SPME does not require any extraction or filtration because any nonvolatile compounds will remain in the headspace vial and will not be adsorbed onto the fiber, yielding a clean sample. This method also helps to eliminate matrix effects due to this being a two-phase system (headspace and fiber) as opposed to a three-phase system (sample, headspace, and fiber) like standard headspace SPME. TV-SPME is like immersion SPME in that immersion SPME is also a two-phase system. With immersion SPME, a fiber is immersed into a liquid (typically aqueous) sample containing the analyte as opposed to extracting the analyte from its vapor. TV-SPME differs from immersion SPME because immersion SPME requires a polar/aqueous matrix in order to generate sufficient driving force for analytes to leave the solution phase and sorb to the fiber coating. In addition, immersion SPME requires much larger sample volumes (e.g., mL).
Many solvents may be used with TV-SPME including methanol, acetone, water, and acetonitrile. SPME fibers should not be exposed to or immersed in chloroform as these solvents can damage the fiber coating. Twenty mL screw cap glass headspace vials have been found to have the best performance with TV-SPME methods. It is recommended to use an autosampler with TV-SPME. While many parameters of the TV-SPME method may be adjusted as desired, the proper volume and extraction temperature must be used for each solvent. Sample volume and extraction temperature are proportional with one another and must be adjusted accordingly. For example, the extraction temperature of a method may be reduced, but the sample volume must also be reduced. This volume may be found by adjusting Equation 3.
Modifications to the derivatization procedure may be made. Derivatization may be performed pre- or post-extraction, at room temperature or heated in the agitator, by exposing the fiber to the vapor of the derivatization agent or by direct immersion of the fiber in the derivatization agent.
There are limitations to the TV-SPME method including the need for compounds to be soluble, thermally stable, and volatile. TV-SPME requires expensive SPME fibers which can have their coating stripped or broken during analysis. These limitations are outweighed by benefits such as large sample volumes relative to typical GC injection volumes, high sensitivity, and no need for filtration. TV-SPME is preferred to headspace SPME because more of the sample is extracted onto the fiber and matrix effects are reduced. TV-SPME is also preferred to immersion SPME because immersion SPME consumes much more sample than TV-SPME. TV-SPME allows for derivatization during the extraction process which reduces analysis time compared to methods such as liquid injection that require that the analyte to be derivatized prior to injection. TV-SPME also requires little to no sample preparation. TV-SPME is simple, efficient, and sensitive for the analysis of a wide variety of samples, including drugs, explosive materials, and racing fuels.
Subscription Required. Please recommend JoVE to your librarian.
Disclosures
The authors have nothing to disclose.
Acknowledgments
This research was supported by the National Institute of Justice (Award No. 2015-DN-BX-K058 & 2018-75-CX-0035). The opinions, findings, and conclusions expressed here are those of the author and do not necessarily reflect those of the funding organizations.
Materials
| Name | Company | Catalog Number | Comments |
| 10 µL Syringe | Gerstel | 100111-014-00 | |
| BSTFA + 1% TMCS (10 x 1 GM) | Regis Technologies Inc. | 50442882 | |
| eVol XR Sample Dispensing System Kit | ThermoFisher Scientific | 66002-024 | |
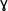 -Butyrolactone (GBL) -Butyrolactone (GBL) |
Sigma-Aldrich | B103608-26G | |
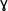 -Hydroxy Butyric Acid (GHB) -Hydroxy Butyric Acid (GHB) |
Cayman Chemicals | 9002506 | |
| Headspace Screw-Thread Vials, 18 mm | Restek | 23083 | |
| Magnetic Screw-Thread Caps, 18 mm | Restek | 23091 | |
| Optima water for HPLC | Fisher Chemical | W71 | |
| SPME Fiber Assembly Polydimethylsiloxane (PDMS) | Supelco | 57341-U | |
| SPME Fiber Assembly Polydimethylsiloxane/Divinylbenzene (PDMS/DVB) | Supelco | 57293-U | |
| Topaz 2.0 mm ID Straight Inlet Liner | Restek | 23313 |
References
- Pawliszyn, J. B. Method and Device for Solid Phase Microextraction and Desorption. United States patent. , (2005).
- Pawliszyn, J. Solid phase microextraction: theory and practice. , John Wiley & Sons. (1997).
- Rainey, C. L., Bors, D. E., Goodpaster, J. V. Design and optimization of a total vaporization technique coupled to solid-phase microextraction. Analytical Chemistry. 86 (22), 11319-11325 (2014).
- Sinnott, R. Chemical Engineering Design: Chemical Engineering. 6, Elsevier. (2005).
- Davis, K. Detection of Illicit Drugs in Various Matrices Via Total Vaporization Solid-Phase Microextraction (TV-SPME). , Indiana University - Purdue University Indianapolis. Master of Science thesis (2019).
- Ash, J., Hickey, L., Goodpaster, J. Formation and identification of novel derivatives of primary amine and zwitterionic drugs. Forensic Chemistry. 10, 37-47 (2018).
- Sauzier, G., Bors, D., Ash, J., Goodpaster, J. V., Lewis, S. W. Optimisation of recovery protocols for double-base smokeless powder residues analysed by total vaporisation (TV) SPME/GC-MS. Talanta. 158, 368-374 (2016).
- Bors, D., Goodpaster, J. Mapping smokeless powder residue on PVC pipe bomb fragments using total vaporization solid phase microextraction. Forensic science international. 276, 71-76 (2017).
- Bors, D., Goodpaster, J. Chemical analysis of racing fuels using total vaporization and gas chromatography mass spectrometry (GC/MS). Analytical Methods. 8 (19), 3899-3902 (2016).
- Beiranvand, M., Ghiasvand, A. Design and optimization of the VA-TV-SPME method for ultrasensitive determination of the PAHs in polluted water. Talanta. 212, 120809 (2020).



