Beer’s Law
Many chemical reactions progress in two directions, forward and reverse. Over time, the forward and reverse reactions will occur at the same rate, and the concentration of the reactants and products will no longer change. This is known as chemical equilibrium.
At chemical equilibrium, the concentrations of each component are related to each other by the equilibrium constant, K, which is the ratio of product concentrations to reactant concentrations, each raised to the power of their stoichiometric coefficients.
But how do you determine equilibrium concentrations? One method measures the intensity of a wavelength of light that the product absorbs before and after it passes through a sample. The intensity difference is called absorbance, and it corresponds to the amount of the absorbing compound in the sample.
You might remember that electrons mostly occupy the ground state. When they absorb a certain amount of energy, they are excited to a higher energy level. That energy corresponds to a specific wavelength of light. You can find this wavelength and measure absorbance with a spectrophotometer, which directs a beam of light through the sample and measures the change in intensity at one or more wavelengths.
The absorbance is equal to the negative log of the intensity of the attenuated light over the intensity of the incident light. By plotting the absorbance values of multiple solutions with different known product concentrations, we observe a linear relationship between absorbance and concentration. This is an example of Beer's Law.
Beer's Law is expressed mathematically by this equation, where A is the absorbance, epsilon is the molar attenuation coefficient, a constant that varies for each compound, l is the path length of light through the sample, and c is the concentration of the compound.
By identifying the linear function for a given compound at a specific wavelength and path length, you can use the absorbance data of a solution at equilibrium to determine the product's equilibrium concentration. From there, you can calculate the equilibrium concentrations of the reactants and solve for the equilibrium constant. In this lab, you will prepare solutions of iron(III) isothiocyanate and use a spectrophotometer to determine its absorbance at various concentrations.
Beer’s Law
Absorbance and Fluorescence
When light hits a substance, it is either absorbed, transmitted, or reflected. Typically, a substance interacts with a range of wavelengths of light, each one interacting with the molecules or atoms differently. A substance may absorb a specific range of wavelengths, reflect another range of wavelengths, and transmit the other wavelengths of light.
When a molecule absorbs light, the energy is used in four different ways: (1) translation, which causes the molecule to change its molecular velocity; (2) vibration, which causes the distance between the molecules to change rapidly; (3) rotation, which causes the atoms to rotate around the bonds in a molecule; and (4) electron excitement, which causes electrons to transition to higher energy levels.
Energy Levels
In 1913, Niels Bohr proposed a model for the hydrogen atom in which electrons travel around the nucleus in fixed, circular orbits, called stationary states. The energy associated with each orbit, or stationary state, exists only at fixed, discrete energies. Only when an electron moves to another orbit is energy absorbed or emitted. The electron is never in-between states. This change only occurs if the absorbed or emitted energy equals the difference between the two energy states.
In Bohr’s model, the quantum number n represents the electron’s energy. When an electron occupies the lowest possible energy state, it is said to occupy the ground state, which is n = 1. When an electron absorbs a photon, whose energy equals the difference between the first and second states, the electron becomes excited and transitions from the ground state to the excited state, where n = 2. If the energy of the photon equals the difference between the first and third states, the electron transitions to the third state, or n = 3, and so on.
Electrons can spontaneously return to the ground state or any other lower, excited state. When this happens, the excess energy that was gained from excitation is released in the form of an emitted photon. The energy of the photon is equal to the difference between the two energy states and corresponds to different wavelengths of light.
Absorption and Emission Spectra
While most substances absorb or emit the maximum amount of light at one wavelength, they also tend to absorb or emit light at a range of wavelengths. This range of wavelengths is called a spectrum. The energy of the absorbed light is quantified and visualized using an absorption spectrum, while the energy of the emitted light is quantified and visualized using an emission spectrum.
Absorption and emission spectra are measured using a spectrophotometer, which is a device that transmits light through a sample and then measures both the wavelength and intensity of light that passes through it. Inside the spectrophotometer is either a diffraction grating or a prism, which separates incoming light into its component wavelengths. The different wavelengths are then transmitted through the sample, and the intensity is recorded on a linear charge-coupled device (CCD) detector. The CCD is an integrated circuit etched onto a silicon surface that forms light-sensitive elements called pixels. The CCD collects and sorts the diffracted light and reads it back at an absorption wavelength.
When measuring the absorbance of a sample, the solute is usually dissolved in a solvent and placed in a container known as a cuvette. Then, the sample is placed inside the spectrophotometer, and the intensity of the transmitted light is measured along with the wavelengths of light to obtain an absorbance spectra. As expected, the intensity of the transmitted light is lower than when there is no sample present inside the spectrophotometer.
This is because the transmitted light is absorbed by the sample, the cuvette, and the solvent. Before measuring samples, the spectrophotometer must be calibrated with a ‘blank’. A blank is a cuvette that only contains the solvent that is used to dissolve the solute. The spectrophotometer is calibrated so that the total absorbance due to the cuvette and solvent is subtracted from the sample’s measured absorbance. This allows us to record the absorbance that is only attributed to the species of interest.
The absorbance is often measured at one wavelength, the maximum absorbance wavelength. However, the absorption can also be measured at a range of wavelengths to acquire the absorption spectrum. For this, the sample is exposed to a range of wavelengths of incident light, and the absorption is recorded at each wavelength. If the sample emits light, the emission spectrum is measured similarly, except the incident wavelength is fixed at the wavelength of maximum absorbance. The instrument then measures the intensity of emitted light over a range of wavelengths.
Beer-Lambert Law
The absorbance of a sample at the wavelength of maximum absorbance provides information about the sample, namely its concentration. The Beer-Lambert Law is an equation that relates transmittance to sample concentration. The transmittance, or intensity of transmitted light, is the fraction of original light that passes through the sample, I, divided by the intensity of the incident light, I0.
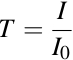
The Beer-Lambert Law states that the optical absorbance, A, of a species in solution is related to the negative log of the transmittance.
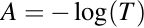
An alternative version of the Beer-Lambert Law states that the optical absorbance, A, of a species in solution is linearly proportional to the concentration, c, of that species when the wavelength, λ, and pathlength, l, are held constant.
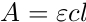
The molar attenuation coefficient, ε, is a measure of how strongly a species absorbs light at a given wavelength. The greater the molar attenuation coefficient, the greater the absorbance. The pathlength, l, is the distance that the light travels through the sample, which is the width of the cuvette. Standard cuvettes have a pathlength of 1 cm.
This linear relationship between absorbance and concentration is a powerful tool that is used to determine the concentration of an unknown sample based on its absorbance. To do this, a standard curve is generated using a gradient of known concentrations of the solute. The absorbance at the peak absorbance wavelength, λmax, is measured for each concentration.
By plotting concentration versus absorbance, a linear relationship is observed that corresponds to the Beer-Lambert equation. The slope of this line is equal to the product of the pathlength and the molar attenuation coefficient. Using this calculated linear function, if the absorbance of the unknown sample is known, the concentration can easily be determined.
If the sample being analyzed is a reaction at equilibrium, Beer’s Law can be used to determine the equilibrium concentration of a product or reactant if the absorbance is measured at λmax specific to that product or reactant. Once the concentration is known, you can determine the equilibrium concentrations of the remaining reactants and products and then solve for the equilibrium constant Keq.
References
- Kotz, J.C., Treichel Jr, P.M., Townsend, J.R. (2012). Chemistry and Chemical Reactivity. Belmont, CA: Brooks/Cole, Cengage Learning.
- Silderberg, M.S. (2009). Chemistry: The Molecular Nature of Matter and Change. Boston, MA: McGraw Hill, Boston.
- Harris, D.C. (2015). Quantitative Chemical Analysis. New York, NY: W.H. Freeman and Company.
