11.14:
固体の構造
11.14:
固体の構造
原子、イオン、または分子が明確な繰り返しパターンで配置されている固体は、結晶性固体として知られています。金属やイオン化合物は、一般的に規則正しい結晶性固体を形成します。結晶性固体は、同じ種類の原子や分子が同じ力やエネルギーで固定されているため、正確な融点を持ちます。非晶質固体または非結晶性固体(場合によってはガラス)で、内部構造に秩序がなく、ランダムに配列されています。大きな分子で構成されている物質や、動きが制限されている分子の混合物は、非晶質の固体を形成することが多いです。非結晶質物質は、分子の構造的な非等価性により、様々な温度範囲で徐々に軟化していきます。非晶質物質を加熱すると、分子間の結合が弱いものから順に壊れていきます。さらに温度を上げていくと、より強い引力が壊れます。
単位セル
結晶性固体の構造は、原子やイオンの位置を表す格子点からなる最も単純な繰り返し単位である単位セルで表現されます。単位セルは、原子やイオンの位置を表す格子点で構成されており、構造全体は、図1に示すように、この単位セルが3次元的に繰り返されることで成り立っています。
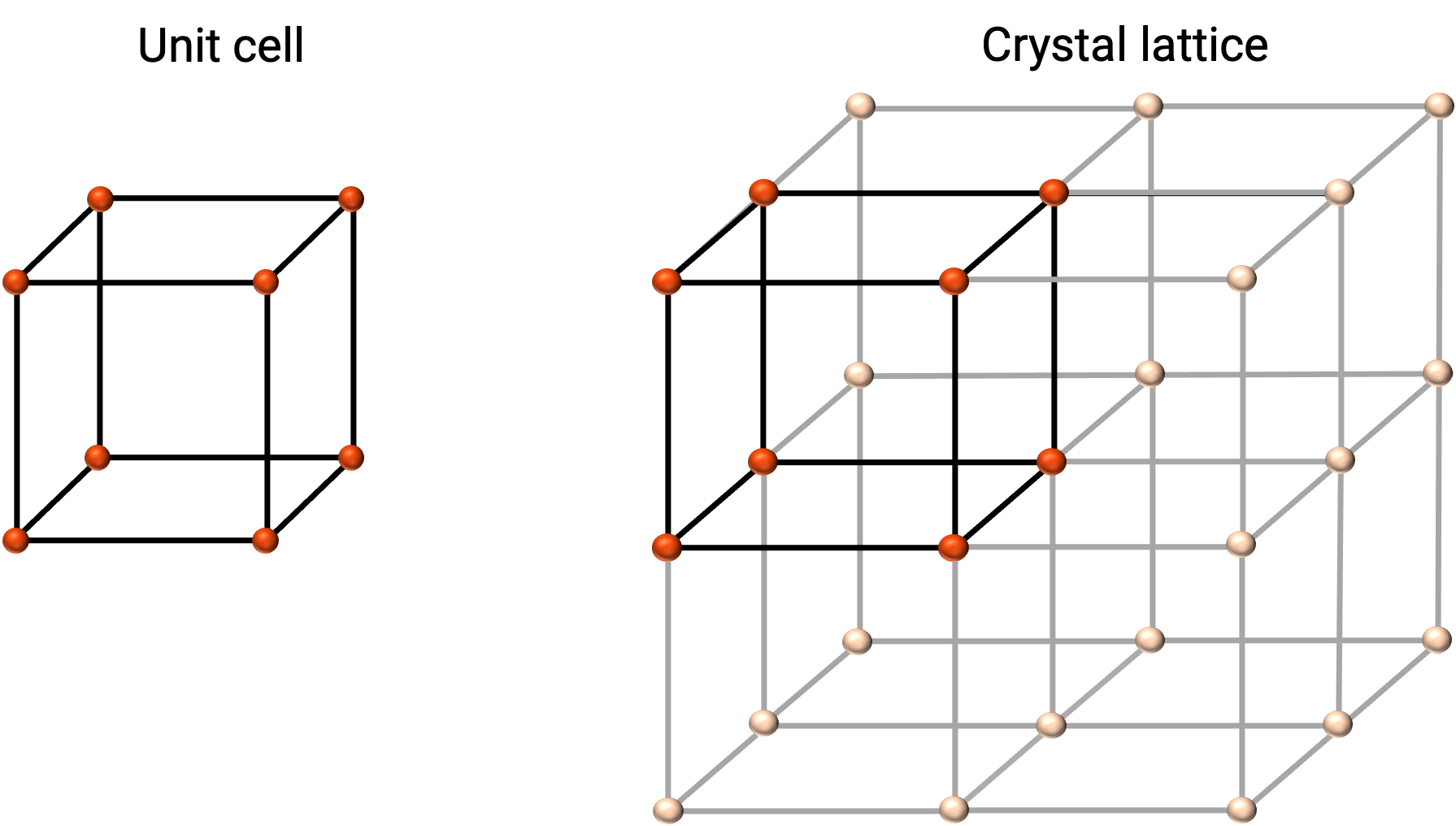
図 1. 単位セルと格子点を赤で示した結晶格子。
一般に、単位セルは、図2に示すように、3つの軸(a、b、c)の長さとそれらの間の角度(α, β, and γ)で定義されます。軸とは、空間格子の点と点の間の長さと定義されます。
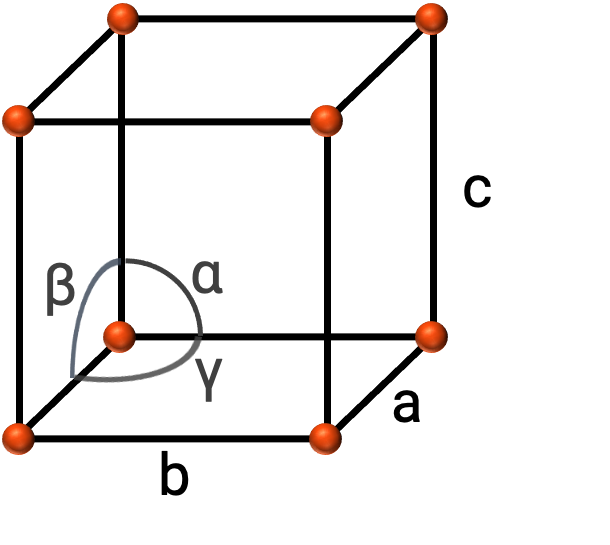
図 2. 単位セルは軸( a 、 b 、 c )と角度( α 、 β 、 および γ )で定義されます。
7種類の格子系があり、中には2種類以上の格子を持つものもあり、合計14種類の単位セルがあります。
| システム | 角度 | 軸 |
| 立方晶 | α = β = γ = 90 ° | a = b = c |
| 正方晶 | α = β = γ = 90 ° | a = b ≠ c |
| 斜方晶 | α = β = γ = 90 ° | a ≠ b ≠ c |
| 単斜晶 | α = γ = 90 ° ; β = 90 ° | a ≠ b ≠ c |
| 三斜晶 | α ≠ β ≠ γ ≠ 90° | a ≠ b ≠ c |
このテキストは、 Openstax 、 Chemistry 2e 、 Section 10.6: Lattice Structures in 結晶固体の構造式から引用したものです。
