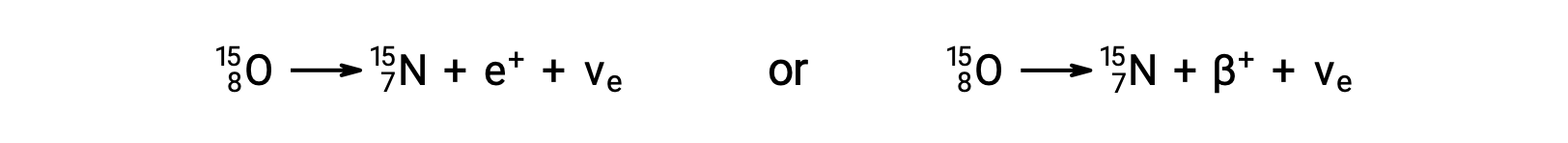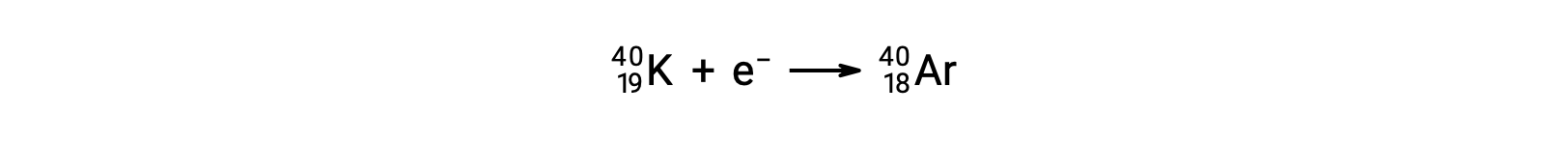Radionuclides disintegrate to daughter nuclides along with the emission of particles or electromagnetic radiation. The fundamental nuclear emissions include alpha particles, beta particles, positrons, neutrons, gamma rays, and X-rays. An alpha particle is composed of two protons and two neutrons, resembling the helium-4 nucleus. Each of these particles has a two-plus charge. Alpha decay reduces the atomic number by two and mass number by four, like the conversion of polonium-210 to lead-206. Beta-minus decay is an emission of high-energy electrons from the nucleus by conversion of a neutron into a proton. The daughter nuclide has an additional proton, and its atomic number is one greater than that of the parent nuclide. During the process, the number of neutrons decreases by one; however, the number of protons increases by one. Thus, the mass number remains unchanged. Beta-plus decay is the conversion of a proton to a neutron, emitting a positively charged particle from the nucleus. This particle has the same mass as an electron, making it an antiparticle of the electron, and is called a positron. The emitted positron reduces the atomic number of a daughter nuclide by one. The positron is short-lived because it quickly collides with an electron, and both particles are annihilated. Their energy is released as two 511 keV gamma rays. Emission of gamma radiation also occurs when an excited daughter nuclide decays to its nuclear ground state. Thus, beta-minus decay of cobalt-60 produces an excited-state nickel-60, which emits two gamma rays while dropping to the nuclear ground state. The mass number and atomic number do not change during gamma decay. The emission of gamma radiation occurs in combination with other nuclear decay reactions. Neutron emission is the ejection of a neutron from the nucleus. It can happen spontaneously, like the decay of beryllium-13 to beryllium-12, or in response to bombardment by gamma rays or particles. The atomic number remains unchanged during this process, whereas the mass number decreases by one. The conversion of potassium-40 to argon-40 exemplifies the emission of energy due to electron capture. The potassium nucleus captures an inner electron in the atom, and a proton converts to a neutron. An outer electron drops to the inner level to fill the vacancy, characterized by an emission of X-rays with an energy corresponding to the transition. The penetration power of alpha particles, which are the most massive of the nuclear particles, is very low, whereas gamma radiation passes through most materials. Neutrons and beta particles can be blocked effectively by relatively lightweight materials.
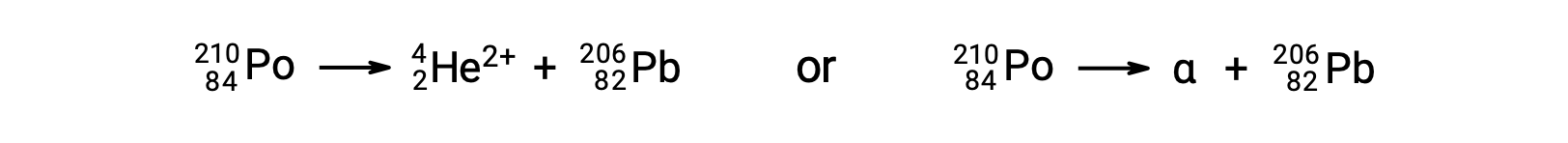
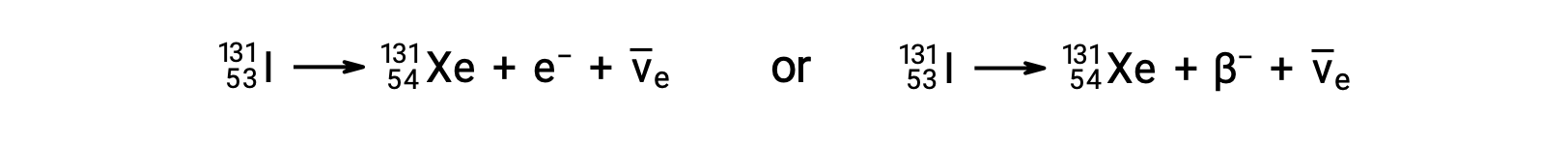
 ) は、エネルギーの保存のために放出されます。
) は、エネルギーの保存のために放出されます。