11.11:
Phase Transitions: Sublimation and Deposition
It is commonly observed that regular ice melts under ambient conditions, but dry ice does not; instead, dry ice transitions directly into the gas phase. This transition from solid to gas — without passing through the liquid phase — is known as sublimation.
Generally, compounds that sublimate exhibit weak intermolecular forces in the solid state. In dry ice, or solid carbon dioxide, weak dispersion forces exist between CO2 molecules.
At atmospheric pressure, dry ice remains a solid below negative 78.5 °C. However, at −78.5 °C, the surface molecules acquire enough thermal energy to completely overcome the attractive forces and transform directly into the vapor phase. This is the sublimation point of dry ice.
The amount of energy required to sublimate one mole of a solid is called its molar heat of sublimation or its molar enthalpy of sublimation. As sublimation is an endothermic process, its enthalpy value is always positive.
The reverse of sublimation — that is, the direct transition from vapor to solid — is called deposition. When gas molecules collide with cooler solid surfaces, they lose heat. Multiple collisions result in a significant loss of heat, and the molecules ultimately deposit.
Since deposition involves loss of energy, it is an exothermic phase change with a negative enthalpy value. Although the enthalpy of deposition is negative, its magnitude is the same as the enthalpy of sublimation.
When sublimation occurs in an open system, most sublimed molecules disperse in air and never come back. Consequently, the rate of sublimation is greater than the rate of deposition.
However, in a closed system, a solid–vapor equilibrium is established at the solid’s sublimation point.
The partial pressure exerted by the gas in dynamic equilibrium with its solid is called its vapor pressure. Solids that sublimate have high vapor pressures. Dry ice, for example, has a vapor pressure as high as 56.5 atm at 20 °C.
However, since most solids have low vapor pressures at easily accessible temperatures, sublimation is not common.
11.11:
Phase Transitions: Sublimation and Deposition
Some solids can transition directly into the gaseous state, bypassing the liquid state, via a process known as sublimation. At room temperature and standard pressure, a piece of dry ice (solid CO2) sublimes, appearing to gradually disappear without ever forming any liquid. Snow and ice sublimate at temperatures below the melting point of water, a slow process that may be accelerated by winds and the reduced atmospheric pressures at high altitudes. When solid iodine is warmed, the solid sublimes and a vivid purple vapor forms. The reverse of sublimation is called deposition, a process in which gaseous substances condense directly in the solid-state, bypassing the liquid state. The formation of frost is an example of deposition.
Like vaporization, the process of sublimation requires an input of energy to overcome intermolecular attractions. Sublimation is, therefore, an endothermic phase transition. The enthalpy of sublimation, ΔHsub, is the energy required to convert one mole of a substance from the solid to the gaseous state. For example, the sublimation of carbon dioxide is represented by:

Likewise, the enthalpy change for the reverse process of deposition is equal in magnitude but opposite in sign to that for sublimation. Because deposition involves the formation of intermolecular forces, it is an exothermic phase transition.

Consider the extent to which intermolecular attractions must be overcome to achieve a given phase transition. Converting a solid into a liquid requires that these attractions be only partially overcome; transition to the gaseous state requires that they be completely overcome. As a result, the enthalpy of fusion for a substance is less than its enthalpy of vaporization. This same logic can be used to derive an approximate relation between the enthalpies of all phase changes for a given substance. Though not an entirely accurate description, sublimation may be conveniently modeled as a sequential two-step process of melting followed by vaporization in order to apply Hess’s Law. Viewed in this manner, the enthalpy of sublimation for a substance may be estimated as the sum of its enthalpies of fusion and vaporization.
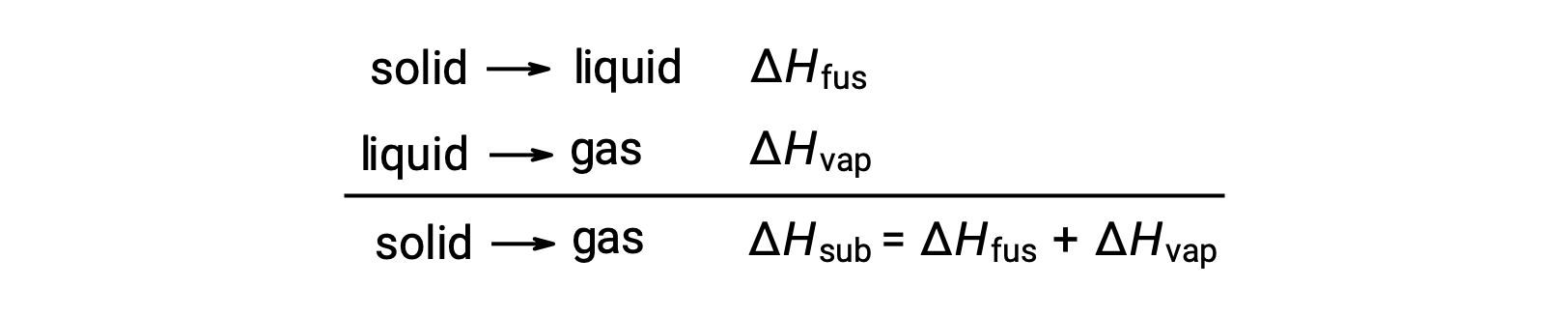
This text is adapted from Openstax, Chemistry 2e, Section 10.3: Phase Transitions.
