10.5:
Structural Isomerism
Isomers are compounds with the same molecular formula but a different structural arrangement. Each isomer has distinct physical and chemical properties. In coordination compounds, one type of isomerism observed is structural isomerism.
In structural isomerism, the connectivities of the atoms to the central metal atom are different despite the same molecular formula. It can be further categorized into coordination-sphere isomers and linkage isomers.
Coordination-sphere isomers occur when the ligand coordinated with the central metal in the coordination sphere interchanges places with a counterion in the lattice. Thus, while the coordination number of the metal ion remains constant, the composition of the coordination-sphere and the compound changes.
In the coordinate complex of cobalt, two coordination-sphere isomers are possible with either bromine as a ligand or chlorine. As the composition of the compound is different, each of these isomers has different chemical properties. When dissolved in water, pentaamminebromocobalt(II) chloride generates chloride ions, while the pentaamminechlorocobalt(II) bromide generates bromide ions in the solution.
Linkage isomerism, on the other hand, occurs when a ligand can coordinate with the central metal atom in two different orientations. Thus, monodentate ligands with more than one potential donor atom are capable of linkage isomerization. Depending on which donor atom coordinates with the metal ion, the name of the ligand changes.
The nitrite ion has two lone pairs of electrons, one on the nitrogen atom and one on the oxygen atom. Either of these can form a coordinate covalent bond with the central metal ion.
When the nitrite ion coordinates through the lone-pair of electrons on the nitrogen atom, it is known as the nitro ligand. When the oxygen atom acts as a donor, it is known as the nitrito ligand.
Coordination complexes of cobalt metal with nitrite ions have two possible linkage isomers, a yellow-colored pentaamminenitrocobalt(III) and an orange-colored pentaamminenitritocobalt(III).
10.5:
Structural Isomerism
Isomerism in Complexes
Isomers are different chemical species that have the same chemical formula. Structural isomerism of coordination compounds can be divided into two subcategories, the linkage isomers and coordination-sphere isomers.
Linkage isomers occur when the coordination compound contains a ligand that can bind to the transition metal center through two different atoms. For example, the CN− ligand can bind through the carbon atom or through the nitrogen atom. Similarly, SCN− can be bound through the sulfur or nitrogen atom. A few different ligands capable of linkage isomerism are shown in Figure 1. The nomenclature of ligands capable of linkage isomerism depends on which donor atom is bonded with the metal ion. For example, a CN− ligand bound through the carbon atom is called cyano, while the same ligand bound through a nitrogen atom is called isocyano.
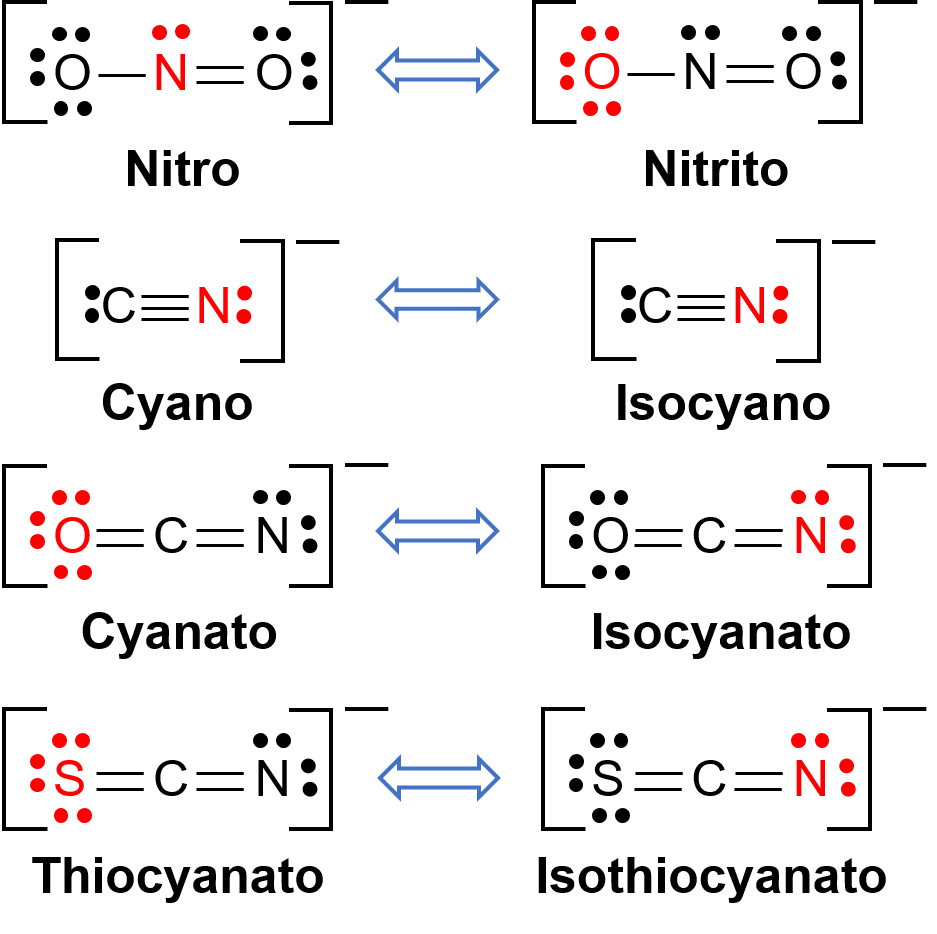
Figure 1. List of different ligands capable of linkage isomerism. In the ligand molecule, the atom in the red binds to the central metal atom.
Coordination-sphere isomers (or ionization isomers) occur when one anionic ligand in the inner coordination sphere is replaced with the counter ion from the outer coordination sphere. A simple example of two coordination-sphere isomers are [CoCl6][Br] and [CoCl5Br][Cl].
This text is adapted from Openstax, Chemistry 2e, Section19.2: Coordination Chemistry of Transition Metals.
