Overview
Source: Laboratories of Jonas T. Kaplan and Sarah I. Gimbel—University of Southern California
Traditional brain imaging techniques using MRI are very good at visualizing the gross structures of the brain. A structural brain image made with MRI provides high contrast of the borders between gray and white matter, and information about the size and shape of brain structures. However, these images do not detail the underlying structure and integrity of white matter networks in the brain, which consist of axon bundles that interconnect local and distant brain regions.
Diffusion MRI uses pulse sequences that are sensitive to the diffusion of water molecules. By measuring the direction of diffusion, it is possible to make inferences about the structure of white matter networks in the brain. Water molecules within an axon are constrained in their movements by the cell membrane; instead of randomly moving in every direction with equal probability (isotropic movement), they are more likely to move in certain directions, in parallel with the axon (anisotropic movement; Figure 1). Therefore, measures of diffusion anisotropy are thought to reflect properties of the white matter such as fiber density, axon thickness, and degree of myelination. One common measure is fractional anisotropy (FA). FA values range from 0, which represents completely isotropic movement, to 1, reflecting maximum anisotropy.
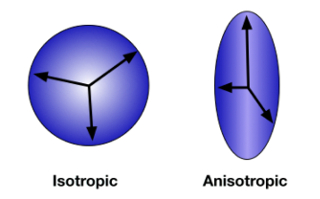
Figure 1: Diffusion anisotropy. When the direction of diffusion is unconstrained and random, movement is measured in all directions equally. This is isotropic diffusion (A). When water molecules are contained within the axon of a neuron, diffusion is anisotropic, tending to occur more frequently along the direction of the axon (B). Please click here to view a larger version of this figure.
In this experiment we will use diffusion tensor imaging (DTI) to measure white matter integrity in traumatic brain injury (TBI). TBI occurs when an external force injures the brain, such as a blow to the head or a sudden movement like the kind that might occur in a car accident. This type of brain injury from mechanical forces is associated with diffuse axonal injury-damage to white matter throughout the brain. Because it is an injury affecting white matter integrity, standard neuroimaging techniques may not reveal the damage. However, measures of diffusion are especially sensitive to these anatomical changes. Following a study by Kraus et al.1, we compare a group of healthy controls to a group of people with TBI and use diffusion imaging to measure the effect of TBI on cerebral white matter. Furthermore, we will test the relationship between white matter integrity and cognitive function using an attention task.2 This study uses a region of interest (ROI) approach focusing on three white matter tracts: the splenium of the corpus callosum, the anterior corona radiata, and the superior longitudinal fasciculus (Figure 2).
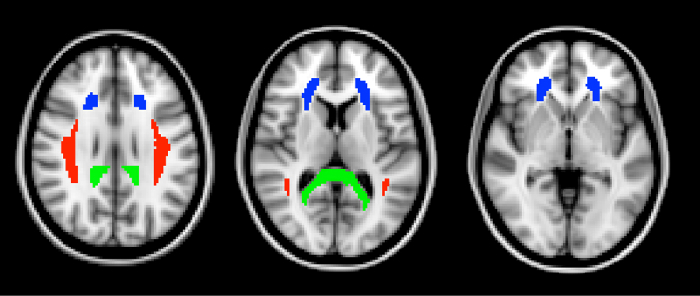
Figure 2: Regions of interest. The three ROIs, defined from the ICBM DTI-81 atlas, are shown here in horizontal slices through the brain. In green is the splenium of the corpus callosum. The splenium is the most posterior part of the corpus callosum. In blue is the anterior corona radiata. The superior longitudinal fasciculus is shown in red. Please click here to view a larger version of this figure.
Procedure
1. Participant recruitment
- Recruit 20 participants with moderate to severe TBI, and 20 age-matched controls. All participants should be over the age of 18.
- TBI patients should have experienced a closed head injury that occurred at least 6 months ago. TBI is diagnosed by assessing several factors such as changes in consciousness, loss of consciousness, and loss of memory from before or after the accident. To be classified as moderate to severe, the patient must have experienced a period of loss of consciousness that was greater than 30 min, and/or obtained a score of less than 13 on the Glasgow Coma Scale.
- Control participants should have no history of neurological or psychological disorders.
- All participants should not have metal in their body. This is an important safety requirement due to the high magnetic field involved in MRI.
- All participants should not suffer from claustrophobia, since the MRI requires lying in the small space of the scanner bore.
- Pre-scan procedures
- Fill out pre-scan paperwork.
- When participants come in for their MRI scan, have them first fill out a metal screen form to make sure they have no counter-indications for MRI, an incidental findings form giving consent for their scan to be looked at by a radiologist, and a consent form detailing the risks and benefits of the study.
- Prepare participants to go in the scanner by removing all metal from their body, including belts, wallets, phones, hair clips, coins, and all jewelry.
- Scanner preparation
- Give the participant ear plugs to protect their ears from the noise of the scanner and ear phones to wear so they can hear the experimenter during the scan, and have them lie down on the bed with their head in the coil.
- Give the participant the emergency squeeze ball and instruct them to squeeze it in case of emergency during the scan.
- Use foam pads to secure the participants head in the coil to avoid excess movement during the scan, and remind the participant that it is very important to stay as still as possible during the scan, as even the smallest movements blur the images.
- Data collection
- Collect a high-resolution T1-weighted anatomical scan. This will be used for registering the participant's brain to the standard atlas space.
- Begin scanning using a pulse sequence optimized for DTI.
- One B0 image is acquired that is not sensitive to diffusion direction.
- Multiple diffusion-weighted images are acquired, each sensitive to a different direction of diffusion. The more directions acquired, the more finely we will be able to resolve the diffusion tensor. However, increasing the number of directions also increases acquisition time. In this study, we will acquire 64 different directions.
- Attention task
- Outside the MRI scanner, have all participants perform a version of the Attention Network Task (ANT)3 to assess their selective attention ability.
- Seat the participant in front of a computer screen, and instruct them on how to complete the task.
- Explain that a series of arrows will appear on the screen. The participant's task is to respond only to the arrow in the center, and to ignore the others. If the central arrow is pointing left, they will press the 'F' key with their left hand. If the central arrow is pointing right, they will press the 'J' key with their right hand. They should respond as quickly and as accurately as possible.
- Begin the task.
- On each trial, present a row of five arrows on the screen. Each arrow can be pointing left or right. On congruent trials, all of the arrows point in the same direction. On incongruent trials, the center arrow points in the opposite direction from the flanking arrows. Each trial begins with a fixation cross that remains on the screen for a variable duration between 400 and 1600 ms. Then the arrow stimuli appear and remain on the screen until the participant responds, or for a maximum of 1700 ms. The trial concludes with a fixation cross that remains on the screen until a total trial duration of 4s has been reached.
- Present 100 trials, half with congruent targets and half incongruent targets.
- Compute the difference in reaction time between incongruent targets and congruent targets. Typically, reaction time is slower in response to incongruent targets. People who are more distracted by the flanking arrows will have a larger difference in reaction time between incongruent and congruent targets. This measure of attentional control will be tested against measures of white matter integrity.
- Post-experiment procedures
- Debrief the participant.
- Pay the participant.
- Data analysis
- Pre-process the diffusion data.
- Visually inspect the data to make sure that it is free from artifacts.
- Perform eddy-current correction with specialized software.
- For each subject, register each of the directional diffusion images to the B0 image using a linear rigid-body affine transformation. This step will compensate for any motion that occurred from scan to scan.
- Remove the skull and other non-brain tissues from the images using automated software. This will ensure that we do not calculate tensors for voxels that are outside the brain.
- Combine among the multiple direction images to calculate the diffusion tensor at each voxel. There are several freely available software packages for processing DTI data that will calculate these values.
- Calculate FA at each voxel, the proportion of tensor magnitude due to anisotropic diffusion.
- Register the diffusion images to the high-resolution anatomical T1 image, and then to the standard atlas space to allow for group-level analysis.
- Define regions of interest (ROI).
- Obtain the three ROI masks from a standard white matter atlas. Here, we use the ICBM-DTI-81 white matter atlas created by the International Consortium for Brain Mapping (Figure 2).
- Register each individual subject's high-resolution anatomical image to the standard atlas.
- Warp the ROI masks into each participant's individual brain space using the registrations performed in the previous step.
- Extract FA values for each subject from each of the three ROIs.
- Compare the FA values between the two groups using Analysis of Variance (ANOVA).
- Compute the Pearson correlation between participants' congruency scores from the ANT and the FA values.
- Pre-process the diffusion data.
Diffusion Tensor Imaging—DTI—is a technique within magnetic resonance that relies on the diffusion of water molecules to study the integrity of white matter networks—the axon bundles that connect cortical and subcortical gray matter—in the brain.
These interconnecting structures can be damaged when an external force, such as a soccer ball, comes into contact with the head during a match. Such repetitive interactions can lead to more severe injury, referred to as Traumatic Brain Injury, TBI for short.
Over time, the damage to cerebral white matter is often associated with deficits in cognition, particularly in the attentional domain. For instance, a player with TBI might be more distracted by a fan in the stands and slower to react to the opposing team.
Based on previous work performed by Kraus and colleagues, this video demonstrates how to measure white matter integrity using DTI in patients with TBI compared to healthy control individuals.
We will also examine cognitive function using a selective attention task, explain how to analyze the imaging data, and interpret the relationship between diffusion values in defined regions of interest and attentional control.
In this experiment, two groups of participants—patients diagnosed with moderate to severe TBI and control individuals that are healthy—are scanned using a DTI protocol, and then tested on an attention task.
First, let’s discuss a few principles behind DTI: The technique is sensitive to the diffusion of water molecules, which are always moving, mostly in parallel to the vast amount of axonal bundles. This type of movement is known as anisotropic diffusion.
Along with the path of water flow, the amount of diffusion can be measured to compute the tensor—essentially illustrated as an ellipsoid with a direction. This value is quantified based on the relative length to its width using a method called fractional anisotropy, abbreviated as FA.
More specifically, FA values can range from 0—isotropic, or equal movement—to 1, which reflects the greatest amount of anisotropy.
The precision is maximized across all voxels by acquiring diffusion images during an MRI scan in many different directions that correspond to shifts in the applied magnetic field. This increased resolution occurs at the expense of time.
Knowing that, a critical dependent variable consists of the FA values for three regions of interest, or ROIs: the anterior corona radiata, the splenium of the corpus callosum, and the superior longitudinal fasciculus.
Compared to normal controls, the FA values are expected to be reduced in individuals with TBI, as their white matter is expected to be compromised in a non-localized manner, resulting in less anisotropy across all ROIs.
However, one region—the anterior corona radiata—is associated with connections to the anterior cingulate cortex, which plays an important role in attentional control.
To further explore this functional relationship between white matter integrity and behavior, all participants are tested on the Attention Network Task developed by Fan and colleagues.
In this paradigm, participants are asked to focus their attention, first towards a fixation cross that appears for a variable duration between 400 and 1600 ms, followed by two different types of arrows: a central one and two flanking groups that may vary in the direction they point.
In half of the trials, all of the arrows point in the same direction; these are considered to be congruent. For the other half, the center arrow points the opposite way of the flanking ones, and these cases are referred to as incongruent. Participants are asked to categorize the arrows by making associated keypresses as quickly as possible when each new set appears.
Here, the difference in reaction times between trial types is calculated as another dependent variable. Individuals who are more distracted by the flanking arrows are expected to have a larger difference score, which indicates poor attentional control.
This measure is therefore predicted to negatively correlate with the FA values, specifically in the region related to attentional ability—the anterior corona radiata—and not the others, thus showing functional significance of particular white matter tracts.
Prior to the experiment, recruit 20 adult patients that have been diagnosed with moderate to severe TBI within the last 6 months and age-matched controls, who have no history of neurological or psychological disorders.
For the purpose of this demonstration, test a patient that’s documented as having a loss of consciousness for more than 30 min or a score of < 13 on the Glasgow Coma Scale.
On the day of their scan, greet them and ensure that they do not suffer from claustrophobia or have any metal in their body; also have them fill out the necessary consent forms detailing the risks and benefits of the study.
After gaining consent, prepare the patient to enter into the scanning room. For more detailed information on the pre-scan procedures, please refer to another MRI project in this collection.
With the patient now in the scanner bore, first collect a high-resolution anatomical scan. Then, initiate a pulse sequence optimized for DTI by acquiring a B0 image—one that is not sensitive to diffusion direction—as well as multiple diffusion-weighted images in 64 different directions.
When the sequence is complete, escort the patient out of the scanner and into an experimental room with a computer to perform the next phase, an Attention Network Task.
Explain the task instructions: They will see a series of five arrows on the screen and should focus only on the center one. To ensure quick and accurate responses, have them place their hands on the keyboard, with their left index finger on the 'F' key and right one on 'J'.
During trials where the center arrow is pointing left, instruct them to press 'F'. Otherwise, when it is facing right, press 'J'.
Allow the patient to complete 100 trials: Remember that each starts with a fixation cross that remains on the screen for a variable duration of 400 to 1600 ms, after which arrows appear, and the central one will either be incongruent or congruent.
Notice that the arrows remain on the screen until the patient responds, or for a maximum of 1700 ms if they don’t press either key. Also note that every trial ends with a fixation cross that remains visible for a total trial duration of 4 s.
At the end of the attention task, debrief the patient to conclude the study, and compensate them for their participation.
After the scanner and behavioral data have been obtained, the raw diffusion files first need to be converted into tensor images.
To begin preprocessing, perform distortion correction for eddy currents, which commonly occur in the raw data as a result of changing the magnetic field during acquisition.
To compensate for motion, apply a linear rigid-body affine transformation, which registers each of the directional diffusion images to the B0 reference.
In addition, isolate the brain from the skull and other non-neural tissues to ensure that tensors are not calculated for voxels outside of the brain.
Now, combine all 64 directional images to calculate the diffusion tensor at each voxel, and output the corresponding FA values in the background.
Subsequently, register the diffusion images to the high-resolution anatomical scan, and then to a standard atlas space to allow for group-level analysis.
With pre-processing completed, use a white matter atlas to identify three ROIs: the anterior corona radiata, the splenium of the corpus callosum, and the superior longitudinal fasciculus.
Finally, register each participant’s anatomical image to the standard atlas, and use these to warp the ROI masks into each participant’s brain space.
To graph the data, compare the extracted FA values by group, plotting each ROI separately. Notice that FA values were significantly lower in the TBI group in all three ROIs, indicating widespread white matter damage in those patients.
To incorporate the behavioral data, plot the differences in reaction times between incongruent and congruent trials against the FA values for all subjects. Perform a Pearson correlation analysis on these overall results, again independently for each ROI.
This time, only one of the regions—the anterior corona radiata—showed a significant correlation, in the negative direction. That is, high FA values were associated with fast reaction times, whereas low anisotropy corresponded to longer response times. These findings suggest a functional link between connecting white matter and attentional control.
Now that you are familiar with how to relate structure and function using DTI and an attentional control task, let’s look at how researchers are using similar approaches to study other populations with compromised connectivity or as a way to protect it.
While normal aging is associated with cognitive decline, changes aren’t necessarily widespread. Rather, they appear to be related to specific functions that correspond to white matter integrity and reduced FA values, especially in the prefrontal cortex. Thus, diffusion imaging could be used as a method for the early detection of age-related changes in executive function.
Using data collected by DTI, researchers can use a 3D technique called fiber tractography and reconstruct white matter tracts throughout the entire brain. This modeling procedure is useful across many domains, including neurosurgery.
If, for instance, surgeons need to remove a tumor, they can plan exactly where the white matter tracts are in proximity to critical structures and avoid detrimental consequences.
You’ve just watched JoVE’s introduction to diffusion tensor imaging. Now you should have a good understanding of how to design and conduct a DTI experiment, as well as how to analyze and interpret specific diffusion patterns with cognitive implications related to traumatic brain injury.
Thanks for watching!
Subscription Required. Please recommend JoVE to your librarian.
Results
The FA values from the three ROIs are shown in Figure 3. Fractional anisotropy was significantly lower in the TBI group in all three ROIs, indicating the presence of widespread white matter damage in those individuals. This non-localized loss of white matter integrity is typical of TBI.

Figure 3: Reduced anisotropy in patients with TBI and relationship with attentional control. (A) FA values are significantly lower in TBI patients compared with healthy controls in all 3 ROIs. (B) FA in the anterior corona radiata correlates negatively with increased incongruency effect in the attention task. Please click here to view a larger version of this figure.
Our measure of attentional control-response time differences between congruent and incongruent targets-correlated negatively with FA values in the anterior corona radiata (Figure 3). In other words, greater differences in response time, indicating poorer attentional control, are associated with decreased FA. These results evidence a relationship between white matter integrity in this location and performance on this task. This relationship was not found in the other two ROIs. The anterior corona radiata is associated with connections to the anterior cingulate cortex, a structure known to play an important part in attentional control.
Subscription Required. Please recommend JoVE to your librarian.
Applications and Summary
Because diffusion imaging can reveal differences in white matter structure that are often not visible with traditional MRI imaging, it is an important tool for understanding brain structure and function. In this experiment we identified a clinically relevant marker for traumatic brain injury that may be used to predict the behavioral consequences of such an injury. DTI has been especially useful in the study of brain development, as changes in white matter structure are found throughout the lifespan from early childhood through late adulthood. For example, aging in older adults is associated with a decline in fractional anisotropy.
More sophisticated analysis of diffusion images allows for the reconstruction and tracing of fiber tracts in the brain, a process known as tractography. Tractography uses the directional information in contiguous voxels to trace specific fiber bundles as they traverse through the brain and can help to build models of the various interconnections among brain structures. This technique can be used to study the connections between individual brain regions of interest, or alternatively to analyze the entire connectome, or complex network structure, of the brain.
Subscription Required. Please recommend JoVE to your librarian.
References
- Kraus, M.F., et al. White matter integrity and cognition in chronic traumatic brain injury: a diffusion tensor imaging study. Brain. 130, 2508-2519 (2007).
- Niogi, S.N., et al. Structural dissociation of attentional control and memory in adults with and without mild traumatic brain injury. Brain. 131, 3209-3221 (2008).
- Fan, J., McCandliss, B.D., Sommer, T., Raz, A., & Posner, M.I. Testing the efficiency and independence of attentional networks. J Cogn Neurosci. 14, 340-347 (2002).




