7.12:
The Pauli Exclusion Principle
Atomic orbitals are the regions where an atom’s electrons are most likely to be found. But how many electrons can each orbital hold?
The Pauli exclusion principle answers this question, as it means that no two electrons in an atom can have the same set of four quantum numbers.
Every orbital corresponds to fixed principal; angular momentum, or azimuthal; and magnetic quantum number values. For example, an electron in the 1s orbital always has a principal quantum number of one and azimuthal and magnetic quantum numbers of zero.
Accordingly, electrons must have different spin quantum number values, or spins, to reside in the same atomic orbital. Recall that the spin quantum number has only two possible values: +1/2 and −1/2. Therefore, only two electrons can occupy the same orbital.
Hence, each s subshell, which has one orbital, can accommodate only two electrons, and each p subshell, which has three orbitals, can hold six electrons. Each of the d and f subshells has a maximum capacity of ten and fourteen electrons, respectively.
The distribution of electrons among an atom’s atomic orbitals is represented by its electron configuration in text or diagram form.
Consider a ground-state hydrogen atom, where one electron occupies the lowest-energy orbital: 1s. The written electron configuration denotes each occupied subshell with the number of the corresponding shell, the letter of the subshell, and a superscript number specifying the number of electrons in the subshell.
The orbital diagram style of the electron configuration denotes each orbital within an occupied subshell as a box or line and each electron as an arrow. An upward arrow indicates a +1/2 spin, or spin-up, and a downward arrow signifies a −1/2 spin, or spin-down. The orbital diagram of hydrogen, therefore, has one upward arrow.
The electron configuration of helium is 1s2. The two electrons have three identical quantum numbers, as they belong to the same shell and subshell. Their spin quantum numbers are different, in accordance with the Pauli exclusion principle.
Electrons with opposite spins are called “paired” if they occupy the same orbital.
For lithium, which has an atomic number of three, the two electrons in the 1s orbital are paired and the electron in the 2s orbital is unpaired. Conventionally, unpaired electrons are represented as spin-up.
7.12:
The Pauli Exclusion Principle
The arrangement of electrons in the orbitals of an atom is called its electron configuration. We describe an electron configuration with a symbol that contains three pieces of information:
- The number of the principal quantum shell, n,
- The letter that designates the orbital type (the subshell, l), and
- A superscript number that designates the number of electrons in that particular subshell.
For example, the notation 2p4 indicates four electrons in a p subshell (l = 1) with a principal quantum number (n) of 2. The notation 3d 8 indicates eight electrons in the d subshell ( l = 2) of the principal shell for which n = 3.
While the three quantum numbers work well for describing electron orbitals, some experiments showed that they were not sufficient to explain all observed results. It was demonstrated in the 1920s that when hydrogen-line spectra are examined at extremely high resolution, some lines are actually not single peaks but, rather, pairs of closely spaced lines. This is the so-called fine structure of the spectrum, and it implies that there are additional small differences in energies of electrons even when they are located in the same orbital. These observations led Samuel Goudsmit and George Uhlenbeck to propose that electrons have a fourth quantum number. They called this the spin quantum number or ms.
In an applied magnetic field, an electron has two possible orientations with different energies, one with spin up, aligned with the magnetic field, and one with spin down, aligned against it.
The fourth quantum number, spin quantum number (ms) describes these two different spin states of an electron. A spin quantum number has two possible values, −1/2 (spin down) and +1/2 (spin-up).
Electron spin describes an intrinsic electron "rotation" or "spinning." Each electron acts as a tiny magnet or a tiny rotating object with angular momentum, or as a loop with an electric current, even though this rotation or current cannot be observed in terms of spatial coordinates.
The magnitude of the overall electron spin can only have one value, and an electron can only “spin” in one of two quantized states. One is termed the α state, with the z component of the spin being in the positive direction of the z-axis. This corresponds to the spin quantum number ms = +1/2. The other is called the β state, with the z component of the spin being negative and ms = −1/2.
Any electron, regardless of the atomic orbital it is located in, can only have one of those two values of the spin quantum number. The energies of electrons having different spins are different if an external magnetic field is applied.
An electron in an atom is completely described by four quantum numbers: n, l, ml, and ms. The first three quantum numbers define the orbital, and are interdependent, while the fourth quantum number is independent of other quantum numbers as it describes an intrinsic electron property called spin. An Austrian physicist Wolfgang Pauli (Nobel Prize in Physics: 1945) formulated a general principle that gives the last piece of information that we need to understand the general behavior of electrons in atoms. The Pauli exclusion principle can be formulated as follows: No two electrons in the same atom can have exactly the same set of all the four quantum numbers. What this means is that two electrons can share the same orbital (the same set of the quantum numbers n, l, and ml) only if their spin quantum numbers have different values. Since the spin quantum number (ms) can only have two values +1/2 and -1/2, no more than two electrons can occupy the same orbital (and if two electrons are located in the same orbital, they must have opposite spins). Therefore, any atomic orbital can be populated by only zero, one, or two electrons.
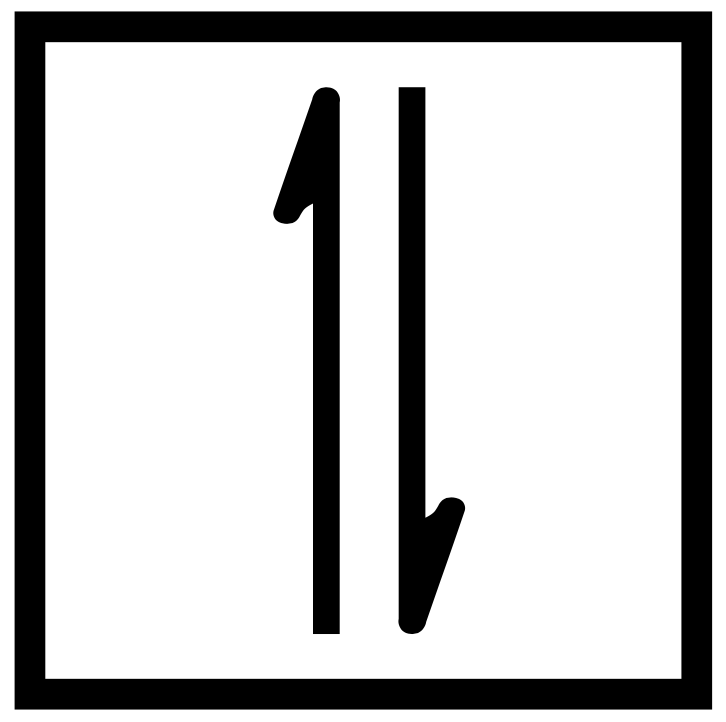
The orbital diagram style of the electron configuration represents each orbital within an occupied subshell as a box or line and each electron as an arrow. The orbital diagram of hydrogen, which an electron configuration of 1s1, is:

An upward arrow indicates a plus-half spin, or spin-up, and a downward arrow signifies a minus-half spin, or spin-down. The orbital diagram of hydrogen, therefore, has one upward arrow.
The electron configuration of helium is 1s2. The two electrons have three identical quantum numbers, as they belong to the same shell and subshell. Their spin quantum numbers are different, in accordance with the Pauli exclusion principle. Electrons with opposite spins are called paired if they occupy the same orbital.
This text is adapted from Openstax, Chemistry 2e, Section 6.3: Development of Quantum Theory.
