Casting Protocols for the Production of Open Cell Aluminum Foams by the Replication Technique and the Effect on Porosity
Summary
Replication is one of the processing techniques used for the production of porous metal sponges. In this paper one implementation of the method for the production of open celled porous aluminum is shown in detail.
Abstract
Metal foams are interesting materials from both a fundamental understanding and practical applications point of view. Uses have been proposed, and in many cases validated experimentally, for light weight or impact energy absorbing structures, as high surface area heat exchangers or electrodes, as implants to the body, and many more. Although great progress has been made in understanding their structure-properties relationships, the large number of different processing techniques, each producing material with different characteristics and structure, means that understanding of the individual effects of all aspects of structure is not complete. The replication process, where molten metal is infiltrated between grains of a removable preform material, allows a markedly high degree of control and has been used to good effect to elucidate some of these relationships. Nevertheless, the process has many steps that are dependent on individual “know-how”, and this paper aims to provide a detailed description of all stages of one embodiment of this processing method, using materials and equipment that would be relatively easy to set up in a research environment. The goal of this protocol and its variants is to produce metal foams in an effective and simple way, giving the possibility to tailor the outcome of the samples by modifying certain steps within the process. By following this, open cell aluminum foams with pore sizes of 1–2.36 mm diameter and 61% to 77% porosity can be obtained.
Introduction
Metal foams have attracted a large amount of interest and research effort in recent years as shown by the large body of work cited in wide ranging review articles such as Banhart1, Conde et al.2 or more recently Goodall and Mortensen3. Among the methods used for production of the material, the replication process is distinguished by its experimental simplicity and the degree of control over the final foam structure that can be offered. It should be noted that although in the literature such materials are often described as foams (and are here) as they are not produced by bubbles of gas within a liquid they are more appropriately called porous metals or microcellular metals.
The first report of the replication process was in the early 1960s4, and it has been developed further at different stages since then, with notable advances by the research group of Mortensen at the Ecole Polytechnique Federale de Lausanne in Switzerland.
The process relies on the casting of the metal around a preform of particles that defines the shape of the porosity in the final material2, 5. After cooling the preform can be removed by solvent leaching or pyrolysis that causes oxidation. A popular use of this technique utilizes NaCl as a space holder to produce aluminum5-10 or aluminum alloy foams11-14. NaCl has several advantages such as being readily accessible, non-toxic and can be removed from the foam by dissolution in water. By having a melting point of 801 °C, it can be used with metals that have a melting point lower than this value, most commonly aluminum, but examples also exist of the use with materials such as bulk metallic glasses, by humidifying a mix of liquid palladium-based bulk metallic glass alloy and NaCl granules15. Substitution of the NaCl with higher melting point materials also permits the production of foams from higher melting point metals16. This may include other water-soluble materials, or insoluble ones including different types of sand. In this form the process becomes more like conventional sand casting as to remove the sand, high pressure water jets17, 18 or different forms of washing19 or agitating20 are required.
The essential process21 proceeds by taking grains of NaCl and placing them in a mold4, 22, 23. The basic method has been used to make aluminum and aluminum alloy foams24-26 for a wide range of foam behavior investigations. Additional steps have been introduced to further control the density and to increase the interconnectivity of the pores; these include the densification of the preform. To densify the preform, sintering has been employed27, 28 and has been used in different experiments since13, with the sintering behavior of NaCl based on temperature, granule size and density described by Goodall et al.29. Another method used for this purpose is cold isostatic pressing (CIP)5, 30; this is a faster technique that can achieve a larger spectrum of comparable densities. The procedure can also be performed in the solid state with metal powder and NaCl grains, and is then sometimes called the Sintering and Dissolution Process31.
A full survey of the use of the replication technique to date and comparison with other techniques is given in Goodall and Mortensen3.
In this work we report in detail equipment and experimental protocols that have been used for the processing of metal foams by the replication method, and which are relatively easy to implement in a research laboratory setting. It is important to acknowledge that other versions of the equipment, with different capabilities exist in other research groups, and that while the equipment presented here is suitable to process the material, it is not the only version or protocol that can be made to work. In any case, a thorough understanding of any particular method is essential for experimental success.
The precise protocols used are detailed below. The protocol variations (A, B, C and D) have small changes between them, principally intended to alter the density of the foams produced. The porosity has been calculated from measurements of the bulk weight of the samples, their volume and the density of aluminum (2.7 g/cm3). In developing the methods described for aluminum foam production by replication, attempts have been made to reduce the amount of advanced equipment to the smallest possible extent, such that the method is as easy to implement as possible. Other variations that may be used at different stages are discussed later.
Protocol
NOTE: The instructions below are for Protocol A (Figure 1). Modifications for Protocol B, C, and D are listed as well.
1. Aluminum Bar Preparation
- Place a large piece (500 g – 1 kg) of commercial purity aluminum ingot into a crucible.
- Place the crucible in a furnace at 800 °C for about 1 hr, until molten.
- Take the crucible out of the furnace and pour the molten aluminum into a cylindrical mold that is 50 mm in diameter, slightly smaller than the final diameter of the chamber to be used for infiltration (51 mm) giving a gap of about ½ mm.
- Wait 1 hr for the bar to cool down.
- Remove the bar from the mold.
- Using a band saw, cut it into 4 same-size pieces.
- Sand the edges of each piece to ensure a good fit in the infiltration mold.
2. Furnace Preparation
- Program the furnace to reach a 740 °C plateau for at least 2 hr.
- Set the heating rate of the furnace to 20 °C/min.
3. Preform Preparation
NOTE: Depending on the height of the foam aimed for, vary the amount of NaCl to use for infiltration between 100 g and 300 g.
- Choose the infiltration NaCl to use, with a diameter corresponding to the pore size range required (for example a range between 1.4 mm and 1.7 mm). The material can be obtained from chemical suppliers in high purity, or supermarket bought table salt can be used (such material will have additives such as iodine and anti-caking agents, but these do not in practice influence the process to a significant extent).
- Select sieves of an appropriate size range and stack on a base container with the smaller opening size at the bottom.
- From the suppliers’ bag of NaCl, take approximately 500 g and pour it in the stacked sieves.
- Agitate the sieves, either manually or using a sieve shaker, for 1 min.
- Discard the NaCl left in the larger aperture size sieve and the bottom container, the NaCl left in the smaller aperture sieve is used for infiltration.
- Weigh the amount of infiltration NaCl obtained.
- If the amount is insufficient, repeat steps 3.4 to 3.7.
NOTE: For Protocols B, C or D, obtain 100 g of fine NaCl (< 500 µm). This creates an extra space in the mold for air trapped in the preform during infiltration in case the air in the preform does not escape the chamber adequately.
4. Mold Preparation
- Using sandpaper and laboratory paper roll, clean the mold cylinder (Figure 2), taking special consideration for both the top and bottom edges, and keeping the mold free of any noticeable impurities from previous use.
- Spray the inside of the mold cylinder with boron nitride aerosol spray, creating a thin coat covering the inside of the mold.
NOTE: This is achieved when the original color of the mold is replaced by a white layer of the spray; it is not necessary to measure its specific concentration. - Let the mold cylinder dry for at least 5 min at RT (heating to around 100 °C for up to 1 hr may be applied for further drying if desired).
- Using fine sandpaper, remove any residue of boron nitride from the edges of the mold cylinder, to improve the seal between the mold cylinder and the mold base.
NOTE: The next 3 steps are for Protocols A and B; for Protocols C and D cut only one gasket ring for the lid. - Cut 2 gasket rings from 1 mm thick graphite sheet (OD = 60 mm, ID = 51 mm), one for the union between the top edge of the mold cylinder and the mold lid leading up to the valve system, the other for the union between the bottom edge of the mold cylinder and the mold base.
- Place one of the gaskets in the mold base groove.
- Place the bottom of the mold cylinder into the groove with the gasket.
- Tap lightly with a mallet on the top of the mold cylinder to secure the bottom to the base groove.
NOTE: For Protocol B, C, or D, add the following step.- Pour the 100 g of fine NaCl (< 500 µm) into the mold cylinder and flatten the top with an uncut aluminum bar tapping the top of it lightly with the mallet to ensure the fine NaCl is packed to a high density.
NOTE: For Protocol D add the following step. - Cut 2 circles of soft 2 mm thick ceramic Kaowool blanket the size of the mold diameter (51 mm) and place them on top of the fine NaCl, use the uncut aluminum bar and the mallet to press them against the fine NaCl.
- Pour the 100 g of fine NaCl (< 500 µm) into the mold cylinder and flatten the top with an uncut aluminum bar tapping the top of it lightly with the mallet to ensure the fine NaCl is packed to a high density.
- Pour the NaCl to be infiltrated into the mold cylinder.
NOTE: For Protocol D add the following step.- Attach the mold and base to a vibrating table, making sure the mold cylinder does not move from the base groove. Vibrate for 1 min at 50 Hz with a 0.01 m amplitude.
- Holding the top of the cylinder in place, pick up the base and shake lightly until the NaCl inside the mold forms a flat surface at the top.
- Place the prepared aluminum bar on top of the NaCl preform.
- Place a graphite gasket in the groove of the mold lid.
- By hand screw the 4 stainless steel studs to the base and secure them with 4 sets of stainless steel nuts and washers on the top of the base using a wrench and place the mold lid on top of the mold cylinder through the studs.
- With a torque wrench set at 16 N·m, screw the 4 sets of steel nuts and washers on the 4 threaded rods screwed into the base and extending up through the lid, where the nuts are tightened to lock the mold lid in place.
- Attach the top of the lid to the valve system with the gasket, clamp, bolt and butterfly nut.
- Close all the valves of the system.
- Open the valve leading to the vacuum pump and the mold (valve 3).
- Turn on the vacuum pump until the dial gauge of the valve system indicates the lowest pressure possible.
- Turn off the vacuum pump.
- If the loss of vacuum in the system is lower than a rate of 50 Torr/sec for the first 10 sec after shutting down the vacuum pump the seal is sufficiently good for infiltration.
- Leave the lid valve open (valve 3) to keep the system at ambient pressure and close the vacuum pump valve (valve 1).
- Without detaching the valve system, place the mold in the preheated furnace and wait for 1 hr.
5. Infiltration
- Close all the valves of the system (Figure 3).
- Open the valve leading to the argon gas cylinder (valve 2).
- Open the main valve on the argon gas tank and set the infiltration pressure with the regulator valve (for a range of 1.4 mm to 1.7 mm of NaCl particle size, use a pressure of 3.5 bars).
NOTE: For Protocol B, an infiltration pressure of 3 bar is used. Use a pressure of 1 bar for Protocols C and D. - In a swift manner, open the lid valve (valve 3).
- After 1 min, remove the mold from the furnace and place it on top of a cooling surface (in this case a copper block).
NOTE: While cooling, the pressure in the system will change. For the first 5 min of this process, pay close attention to the pressure indicated by the regulator and adjust back to the infiltration pressure if necessary.
6. Sample Extraction
- After 30 min, when the mold is cool enough to handle with light heat resistant gloves, detach the valve system and place the mold base on a workbench vise. Unscrew the lid from the top of the cylinder.
- With the lid off, lightly tap the top of the mold cylinder with a mallet in a perpendicular direction to the vise’s grip to loosen the mold cylinder from the base groove.
- Remove the mold base from the vise and place the mold cylinder in the vise grip.
- With the mallet tap the remaining aluminum on top of the sample to push it out of the mold cylinder.
- Using a band saw, cut the bottom part of the foam sample, removing the surplus aluminum.
- Depending on the height of foam required, cut where desired, close to the top of the sample.
- Place the infiltrated foam in a beaker with water and a magnetic stirring bar on a stirring hot plate to dissolve the NaCl preform.
- Set the temperature of the hot plate to 60 °C. Change the water every 10 min until there is no NaCl left in the foam.
NOTE: To ensure there is no NaCl left in the foam, change the water approximately 10 times. It is also possible to make periodic checks of the sample weight after a brief drying stage. When this ceases to change significantly with further immersion, the NaCl must be completely removed. - Finally using an electric air drier remove all the water left in the foam. The foam sample is ready.
Representative Results
In Figure 4 the morphology of the NaCl grains can be seen (angular and spherical), for illustrative purposes. The foams obtained with Protocol A were made using angular shaped grains and the rest were made with the spherical grains. It was found that the use of different shape NaCl grains had no observed effect on the porosity obtained in the samples.
From the results we can determine that samples a, b, and c (made with Protocol A), are on average 63% porous (Figure 5), determined from their bulk weight and volume. By making changes to the technique, for example including the pocket of fine NaCl at the bottom, enables the process to produce foams 5% more porous and allows the infiltration pressure to be lowered (from 3.5 to 3 bar), these are samples d, e and f made with Protocol B (Figure 6). The only difference between Protocols A and B is the addition of the fine NaCl at the bottom of the preform.
By removing the bottom gasket of the infiltration mold, as is done in Protocol C (Figure 7), the required infiltration pressure may be reduced further (from 3 to 1 bar). Using this method, samples g, h and i were produced, also showing a 5% increase in porosity. In Protocol C the reason for using 3 different sizes of NaCl particles is to investigate any effect on porosity, and demonstrate that, even with this change, the porosity obtained in the foams remains very similar and the change in particle size has little to no effect on foam porosity when compared to the effect of the protocol used. The foams produced with Protocol C are three separate samples, each one made with a different particle size. The final set of samples, j, k and l were made using Protocol D (Figure 8), by vibrating the NaCl to be infiltrated, increasing the density of the preform, giving a large jump of 8% in the porosity of the foams.
Occasional observations of an unsuccessful infiltration are that a certain region or regions of the preform are not properly infiltrated; over infiltration might occur as well, such as encapsulation of several NaCl particles by the metal, caused mainly by a high infiltration pressure, preventing the water to leach the NaCl out; this is very evident when there is a large drop in the apparent porosity (greater than 5%) in a sample produced using a certain Protocol, though this is a very rare event. Figure 9 shows a non-infiltrated sample on the left, a properly infiltrated sample in the middle and an over infiltrated sample on the right. In Figure 10 the change in porosity by modifying the infiltration pressure can be seen. If a higher infiltration pressure is applied, more aluminum is forced between the NaCl spheres (the higher pressure allows the surface tension to be overcome to a greater degree, permitting narrower gaps to be filled with metal), thus the free space remaining decreases, decreasing the porosity. To control the outcome of a properly infiltrated sample by this method is more difficult when compared to using a different protocol, since at higher pressures the risk of blocked pores in the foam increases greatly.
To assess the outcome of a production run the first indicator of properly infiltrated foams is their density, another would be observing the outside of the sample; a completely infiltrated foam is uniform in all its structure, if there are errors, they are quite noticeable (mostly blocked pores or non-infiltrated zones); they can be seen in Figure 11. The final results of this procedure are indicated in Table 1.

Figure 1. Foam Replication General Protocol Steps.
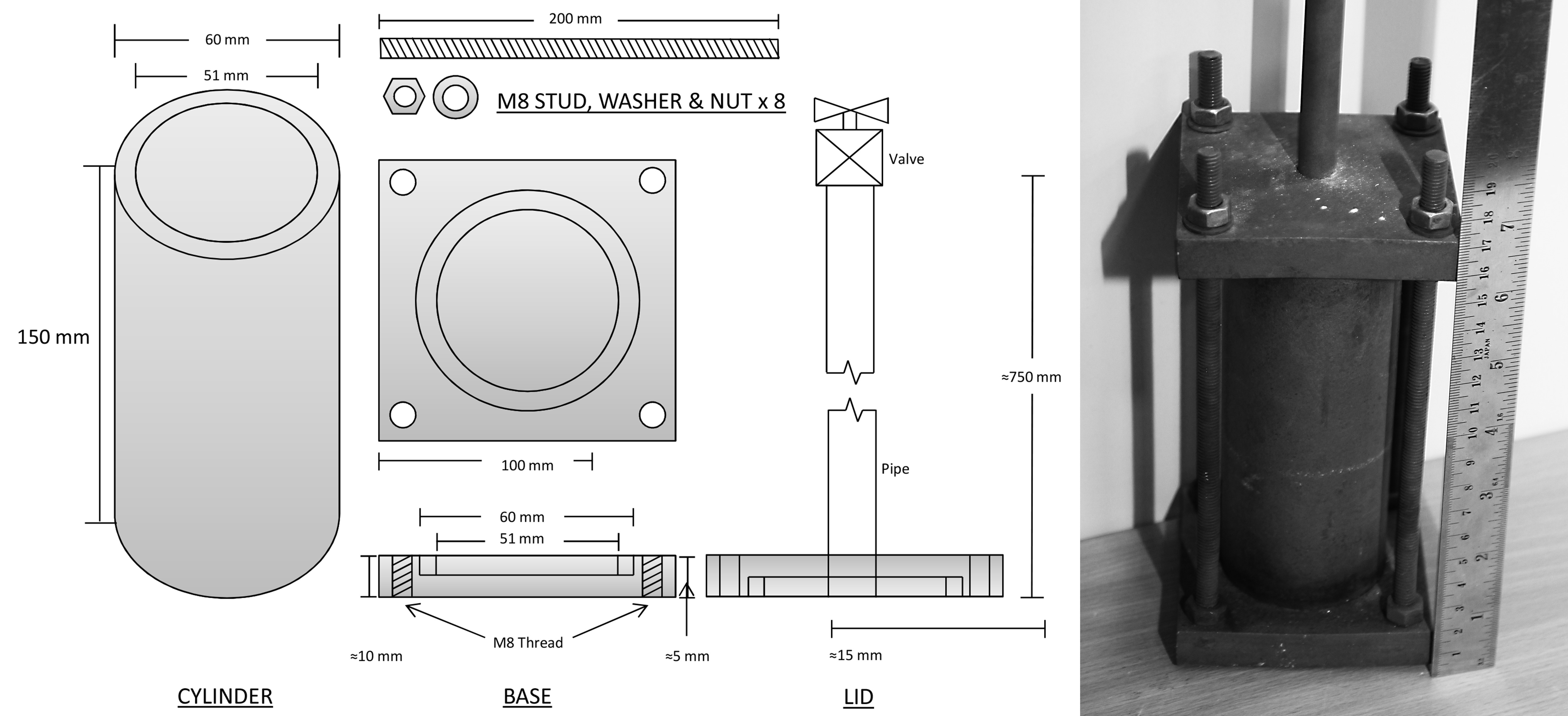
Figure 2. Design Schematics of the Foam Infiltration Mold and Assembled Image (Metric Scale). Please click here to view a larger version of this figure.

Figure 3. Laboratory Schematics of the Foam Infiltration Rig.

Figure 4. Morphology of the NaCl Grains (Left: Angular 2–2.36 mm; Right: Spherical 1.4–1.7 mm).

Figure 5. Protocol A samples a, b and c are made of open porous 99.95% aluminum foam with a pore size range from 1.4 mm to 1.7 mm, an average porosity of 63%, measuring 51 mm in diameter and 25.4 mm in height (Metric Scale). Please click here to view a larger version of this figure.

Figure 6. Protocol B samples d, e and f are made of open porous 99.95% aluminum foam with a pore size range from 1.4 mm to 1.7 mm, an average porosity of 66%, measuring 51 mm in diameter and 25.4 mm in height (Metric Scale). Please click here to view a larger version of this figure.

Figure 7. Protocol C samples g, h and i are made of open porous 99.95% aluminum foam with a pore size range of 1 mm to 1.18 mm, 1.4 mm to 1.7 mm and 2 mm to 2.36 mm respectively, an average porosity of 70%, measuring 51 mm in diameter and 25.4 mm in height (Metric Scale). Please click here to view a larger version of this figure.

Figure 8. Protocol D samples j, k and l are made of open porous 99.95% aluminum foam with a pore size range of 1.4 mm to 1.7 mm, an average porosity of 76%, measuring 51 mm in diameter and 25.4 mm in height (Metric Scale). Please click here to view a larger version of this figure.

Figure 9. Effect of the Infiltration Pressure on the Foams (Left: Non-Infiltration; Middle: Correct Infiltration; Right: Over Infiltration) (Metric Scale). Please click here to view a larger version of this figure.

Figure 10. Replicated Foam Porosity Variation by Infiltration Pressure Change using only Protocol A.

Figure 11. Noticeable Errors in Foams Produced by this Method (Left: Transversal Image; Right: Side Image) (Metric Scale). Please click here to view a larger version of this figure.
| Protocol | Sample | Particle Size (mm) | Porosity (%) |
| A | a | 1.4 – 1.7 | 63.45 |
| A | b | 1.4 – 1.7 | 62.98 |
| A | c | 1.4 – 1.7 | 63.09 |
| B | d | 1.4 – 1.7 | 66.33 |
| B | e | 1.4 – 1.7 | 66.21 |
| B | f | 1.4 – 1.7 | 66.08 |
| C | g | 1 – 1.18 | 69.96 |
| C | h | 1.4 – 1.7 | 70.03 |
| C | i | 2 – 2.36 | 70.75 |
| D | j | 1.4 – 1.7 | 76.20 |
| D | k | 1.4 – 1.7 | 75.69 |
| D | l | 1.4 – 1.7 | 76.56 |
Table 1. Replicated foam sample characteristics, porosities obtained and preform size used.
Discussion
The basic method described here has been used in different forms by other researchers. Some of the key variants that allow foams of different types to be created are discussed. In characterizing these foams we have measured the porosity, as this is a quick and easy assessment to make, but characterization of other structural characteristics, such as pore size, specific surface area or strut thickness might be required to obtain a full understanding of foam characteristics for different applications. In practice, for production of foams by replication, the pore size is well-controlled by the particle size of the NaCl used, and links between this, the density and the other structural characteristics can be made.
Densified preforms
In this present contribution we have described a NaCl preform that is made by tipping NaCl grains into a chamber. While, as discussed, some degree of control over the density can be achieved by vibrating the sample, the range accessible remains rather restricted, due to the limited range of packing fractions of the NaCl that can be achieved. In order to produce foams of higher porosity, the density of the preform can be increased by compacting it mechanically (for example in Cold Isostatic Pressing to preserve isotropic structure), or by sintering where densification is driven by surface area reduction. Both of these methods would be expected to be more effective for smaller NaCl particle size (sub millimeter), as smaller grains are less liable to crack and have a larger surface area to volume ratio. As the pore size used in the experiments described in this paper is larger, and either process would require additional equipment, with the focus on a simple and easily implemented process, they have not been used.
Shaped preforms
In Goodall and Mortensen14 a method is introduced to control the pore size and shape further than is possible using single NaCl grains. In this method fine NaCl powder is mixed with a binder (for simplicity, flour and water can be used), then shaped into the desired form before a heat treatment is used to thermally remove the binder. Although not experimentally complex, this method has not been used in our experiments as it is not essential to produce a foam and requires somewhat more precise pressure control to ensure the fine scale porosity in the preform itself is not infiltrated.
Alternative preforms to NaCl
Although NaCl displays many desirable features as a preform material (including relatively high melting temperature, high degree of solubility in water and low toxicity and cost), it is not always suitable. One particular case is when higher melting point metals are to be processed, and in this situation it can be replaced with other materials, such as sodium aluminate16. These materials improve the temperature capability, but are generally more expensive and challenging to dissolve, and are not required to process foams from relatively low melting point metals, such as aluminum, the most common metal from which foams are made.
Cold wall / partial cold wall pressure chambers
To infiltrate metal into finer particle size preforms, to produce smaller pore size foams, higher pressures will be required. The test rig described in this work is suitable for use up to 6 atm pressure, but as the pressure is increased the probability of leaks from the seals goes up. This can be solved by alternative designs of pressure chamber, where the sealed regions are separated from the heated zone, usually protected by cooling water. While the capability of such equipment is increased over that described here, the design and fabrication is significantly more complex, and so has not been implemented in this version.
Characteristics of this embodiment of the process
While casting the molten aluminum into the bar mold, a pipe defect will form at the top, due to solidification shrinkage. The best results are obtained with fully solid ingots, so this part should be discarded or recycled.
It has been found that in order for the process to work, the seal between the molten metal and the mold wall must be good (otherwise argon will bypass the metal and no infiltration will occur). For this reason when applying infiltration pressures of 3 bars or higher, the best results are obtained with a large amount of aluminum, sufficient to fill the mold, even if the aim is obtaining short foams, as this increases the pressure of the liquid metal around the mold at the top of the preform and improves the seal. A small gap of half a centimeter has been found to be the ideal height between the aluminum piece and the mold lid for the current equipment. For infiltration pressures of 2.5 bars or less the size of the gap is irrelevant, the only amount of aluminum needed is the enough to completely fill the preform.
When tightening the nuts on the studs use a star pattern (tightening opposite pairs in a stepwise fashion) to ensure that the pressure around the gasket is even and a seal is obtained. To avoid damage to the valves on closing, this is always done manually.
Occasionally, there can be defects or regions of poor infiltration. These are most likely to form at the bottom, where the molten metal has furthest travel, or at the top, near the interface with the dense metal. Therefore the most consistent part of the sample is in the center of the region occupied by the NaCl preform. The top and bottom parts of the foam may be cut away and discarded. Whenever it is required to cut the foam to produce a sample, it is best to do so with the NaCl still present in it. If the cuts are made after leaching, wherever the cut is made, it will damage and block the structure of the foam. Where cutting the sample after leaching is required, a successful method is to use a non-loading technique such as Electro-Discharge Machining (EDM, also called spark erosion).
There are many variables in the process that can be modified for different effect, but in order to change the porosity the most suitable control variables are either the preform density or the infiltration pressure used.
The purpose of using different protocols (A, B, C and D) is to produce foams with different porosities, from 61% to 77%. Applying Protocol A will produce samples with 63% porosity on average; Protocol B produces samples with 66% porosity; Protocol C produces samples with 70% porosity and Protocol D produces samples with 76% porosity. By adding the fine NaCl at the bottom of the mold in Protocols B, C and D it creates a refuge for air trapped in the preform during infiltration if the evacuation of the chamber is not perfect. The NaCl being much finer will resist infiltration by the aluminum until higher pressures are reached, ensuring that the preform is completely infiltrated. Without this any air present will be compressed, not eliminated and unwanted additional porosity will be present, most likely as uninfiltrated regions. Protocols C and D were developed to allow infiltration to be achieved with much lower pressures. For the samples represented in Figure 6 a different particle size preform was used, it can be noted that this change does not have a significant effect when compared to the protocol used.
By not using the bottom gasket in Protocols C and D a small flow of gas through the bottom of the mold is possible, meaning that trapped gas from the preform can be evacuated without having to be compressed to higher pressures. If this was done without the fine NaCl then aluminum may also be forced out, but as this layer resists penetration by the liquid aluminum at the pressures applied it will prevent aluminum escape.
In Protocol D, by vibrating the preform, a higher porosity foam can be achieved; roughly 9–10% more porous when compared to Protocol C. This happens since the NaCl grains in the preform are closer together, leaving less space to be filled by the aluminum. The ceramic sheet is added in Protocol D to prevent the fine NaCl to mix with the infiltration NaCl during vibration, no significant effect was found in the final products when adding the ceramic sheet to Protocol C.
The principal limitation for the foam processing technique described is the porosity of the foams; the lowest achieved so far with the rig and protocols described here are around 61% and the highest close to 77%. However it is a cheap and easy to use technique compared to more complicated and expensive methods such as investment casting, sintering or additive manufacturing. Another limitation is the metals that can be used; any metal having a melting point too close or above the NaCl melting point (801 °C) cannot be infiltrated with this preform. Aluminum, magnesium and tin have been processed using this technique.
Equipment and several successful protocols for the production of aluminum foams are presented in detail. Using this method it is possible to create aluminum open celled foams with porosities of 61–77% (corresponding to density in the range of 1,053 to 621 kg/m3) and with pore sizes in the range 1 to 2.36 mm diameter. Furthermore, it is known that with variations in the conditions used, some of them relatively minor, these ranges can be extended significantly, and other variables, such as pore shape can be changed. The replication technique is highly suitable for research laboratory use for metal foam production.
Disclosures
The authors have nothing to disclose.
Acknowledgements
The corresponding author would like to acknowledge the Mexican Government’s National Council of Science and Technology CONACYT for provision of a scholarship.
Materials
| Name of Reagent/ Equipment | Company | Catalog Number | Comments/Description |
| Salt | Hydrosoft | Granular Salt 25 kg 855754 | http://www.travisperkins.co.uk/p/hydrosoft-granular-salt-25kg/855754/3893446 |
| Aluminum | William Rowland | Aluminum Ingots 99.87% pure 25 kg drum | http://www.william-rowland.com/products/high-purity-metals#product-id-1 |
| Crucible | Morgan Advance Materials | Syncarb Crucible | http://www.morganmms.com/crucibles-foundry-products/crucibles/syncarb/ |
| Furnace | Elite Thermal Systems | TLCF10/27-3216CP & 2116 O/T | http://www.elitefurnaces.com/eng/products/furnaces/1200%20Top%20Loading%20Furnaces.php |
| Bar Mold | The University of Sheffield | Custom Made | Stainless Steel 304, 15 cm height, 5 cm inner diameter, 6 cm outer diameter |
| Band Saw | Clarke | CBS45MD (6" x 4 1/2") 370W 060710025 | http://www.machinemart.co.uk/shop/product/details/cbs45md-41-2in-x-6in-metal-cutting-ban |
| Sandpaper | Wickes | Specialist wet & dry sandpaper 501885 | http://www.wickes.co.uk/Specialist-Wet+Dry-Sandpaper-PK4/p/501885 |
| Sieves | Fisher Scientific | Fisherbrand test sieves 200 mm diamater | http://www.fisher.co.uk/product/brand_listing.php/F/Fisherbrand/Sieve |
| Balance | Precisa | XB 6200C | http://www.precisa.co.uk/precision_balances.php |
| Boron Nitride | Kennametal | 500 ml spray can | http://www.kennametal.com/content/dam/kennametal/kennametal/common/Resources/Catalogs-Literature/Advanced%20Materials%20and%20Wear%20Components/B-13-03401_ceramic_powders _brochure_EN.pdf |
| Infiltration Mold, Base and Lid | The University of Sheffield | Custom Made | Stainless Steel 304, 15 cm height, 5.1 cm inner diameter, 6 cm outer diameter |
| Cylindrical Mold | The University of Sheffield | Custom Made | Low carbon steel 1020, 15 cm height, 5 cm inner diameter, 6 cm outer diameter |
| Graphite Gasket | Gee Graphite | Geegraf Stainless Steel Reinforced Graphite 1 mm thick | http://www.geegraphite.com/steel_reinforced.html |
| Mallet | Thor Hammer Co. Ltd. | Round Solid Super Plastic Mallet | http://www.thorhammer.com/Mallets/Round/ |
| Wrench | Kennedy Professional | 13 mm Ratchet Combination Wrench KEN5822166K | https://www.cromwell.co.uk/KEN5822166K |
| Nuts | Matlock | M8 Steel hex full nut galvanized | https://www.cromwell.co.uk/CTL6400068J |
| Washers | Matlock | M8 Form-A steel washer bzp | https://www.cromwell.co.uk/CTL6451208H |
| SS Nuts | Matlock | M8 A2 st/st hex full nut | https://www.cromwell.co.uk/CTL6423008F |
| SS Washers | Matlock | M8 A2 st/st Form-A washer | https://www.cromwell.co.uk/CTL6464008H |
| Stainless Steel Studding | Cromwell | M8 x 1 Mtr A2 Stainless Steel Studding QFT6397080K | https://www.cromwell.co.uk/QFT6397080K |
| Valves | Edwards | C33205000 SP16K, Nitrile Diaphragm | https://www.edwardsvacuum.com/Products/View.aspx?sku=C33205000 |
| Fitting Cross | Edwards | C10512412 NW16 Cross Piece Aluminum | https://www.edwardsvacuum.com/Products/C10512412/View.aspx |
| Fitting T | Edwards | C10512411 NW16 T-Piece Aluminum | https://www.edwardsvacuum.com/Products/C10512411/View.aspx |
| Vacuum Pump | Edwards | A36310940 E2M18 200-230/380-415V,3-ph, 50Hz | http://www.edwardsvacuum.com/Products/View.aspx?sku=A36310940 |
| Dial Gauge | Edwards | D35610000 CG16K, 0-1040mbar | http://www.edwardsvacuum.com/Products/View.aspx?sku=D35610000 |
| Argon Gas | BOC | Pureshield Argon Gas | http://www.boconline.co.uk/en/products-and-supply/industrial-gases/inert-gases/pureshield-argon/pureshield-argon.html |
| Stainless Steel Hose | BOC | Stainless Steel Hose | http://www.boconline.co.uk/en/products-and-supply/speciality-equipment/hoses-and-pigtails/index.html |
| Regulator | BOC | HP 1500 Series Regulator | http://www.boconline.co.uk/en/products-and-supply/speciality-equipment/regulators/single-stage-regulators/hp1500-series/hp1500-series.html |
| Copper Block | William Rowland | Copper Ingot 25 kg | http://www.william-rowland.com/products/high-purity-metals#product-id-18 |
| Vise | Record | T84-34 H/Duty Eng Vice 4 1/2" Jaws REC5658326K | https://www.cromwell.co.uk/REC5658326K |
| Beaker | Fisher Scientific | 11567402 – Beaker, squat form, with graduations and spout 800mL | https://webshop.fishersci.com/insight2_uk/getProduct.do;jsessionid=16D5812 D71B8CB37B475E94281E2BEA 5.ukhigjavappp11?productCode=11567402&resultSet Position=0 |
| Stirring Hot Plate | Corning | Corning stirring hot plate Model 6798-420d | http://www.corning.com/lifesciences/us_canada/en/technical_resources/product_guid/shp/shp.aspx |
| Stir Bar | Fisher Scientific | 11848862 – PTFE Stir bar + Ring 25×6 mm | https://webshop.fishersci.com/insight2_uk/getProduct.do;jsessionid=16D5812 D71B8CB37B475E94281E2BEA 5.ukhigjavappp11?productCode=11848862&resultSet Position=0 |
| Air dryer | V05 | V05 Max Air Turbo Dryer DR-120-GB | http://reviews.boots.com/2111-en_gb/1120627/v05-v05-max-air-turbo-hair-dryer-dr-120-gb-reviews/reviews.htm |
| Ceramic Sheet | Morgan Advance Materials | Kaowool Blanket 2 mm thick | http://www.morganthermalceramics.com/downloads/datasheets?f[0]=field_type%3A84 |
| Vibrating Table | Pevco | Pevco Vibrating Table 1.25m x 0.625m x 0.6m | http://www.peverilmachinery.co.uk/equipment/vibrating-tables |
References
- Banhart, J. Manufacture, characterisation and application of cellular metals and metal foams. Progress in Materials Science. 46, 559-632 (2000).
- Conde, Y., Despois, J. -. F., Goodall, R., Marmottant, A., Salvo, L., San Marchi, C., Mortensen, A. Replication processing of highly porous materials. Advanced Engineering Materials. 8 (9), 795-803 (2006).
- Goodall, R., Mortensen, A., Laughlin, D. E., Hono, K. Chapter 24. Porous Metals. Physical Metallurgy. , 2399-2595 (2014).
- Polonsky, L., Lipson, S., Markus, H. Lightweight Cellular Metal. Modern Castings. 39, 57-71 (1961).
- San Marchi, C., Mortensen, A., Degischer, H. P., Kriszt, B. Chapter 2.06. Infiltration and the Replication Process for Producing Metal Sponges. Handbook of Cellular Metals. , 44-56 (2002).
- Galliard, C., Despois, J. F., Mortensen, A. Processing of NaCl powders of controlled size and shape for the microstructural tailoring of aluminium foams. Materials Science and Engineering A. 374 (1-2), 250-262 (2004).
- Despois, J. F., Mortensen, A. Permeability of open-pore microcellular materials. Acta Materialia. 53 (5), 1381-1388 (2005).
- Goodall, R., Despois, J. F., Marmottant, A., Salvo, L., Mortensen, A. The effect of preform processing on replicated aluminium foam structure and mechanical properties. Scripta Materialia. 54, 2069-2073 (2006).
- Goodall, R., Marmottant, A., Salvo, L., Mortensen, A. Spherical pore replicated microcellular aluminium: Processing and influence on properties. Materials Science and Engineering A. 465 (1-2), 124-135 (2007).
- Despois, J. F., Marmottant, A., Salvo, L., Mortensen, A. Influence of the infiltration pressure on the structure and properties of replicated aluminium foams. Materials Science and Engineering A. 462, 68-75 (2007).
- San Marchi, ., Despois, C., F, J., Mortensen, A. Uniaxial deformation of open-cell aluminium foam: the role of internal damage. Acta Materialia. 52 (10), 2895-2902 (2004).
- Goodall, R., Weber, L., Mortensen, A. The electrical conductivity of microcellular metals. Journal of Applied Physics. 100, 044912 (2006).
- Kadar, C., Chmelik, F., Kendvai, J., Voros, G., Rajkovits, Z. Acoustic emission of metal foams during tension. Materials Science and Engineering A. 462, 316-319 (2007).
- Goodall, R., Mortensen, A. Microcellular aluminium. Child’s Play! Advanced Engineering Materials. 9 (11), 951-954 (2007).
- Wada, T., Inoue, A. Fabrication, Thermal Stability and Mechanical Properties of Porous Bulk Glassy Pd-Cu-Ni-P Alloy. Materials Transactions. 44 (10), 2228-2231 (2003).
- DeFouw, J. D., Dunand, D. C. Processing and compressive creep of cast replicated IN792 Ni-base superalloy foams. Materials Science & Engineering A. 558, 129-133 (2012).
- Berchem, K., Mohr, U., Bleck, W. Controlling the Degree of Pore Opening of Metal Sponges, Prepared by the Infiltration Preparation Method. Materials Science and Engineering A. 323 (1-2), 52-57 (2002).
- Lu, T. J., Ong, J. M. Characterization of closed-celled cellular aluminum alloys. J. Mater. Sci. 36, 2773-2786 (2001).
- Chou, K. S., Song, M. A. A Novel Method for Making Open-cell Aluminum Foams with Soft Ceramic Balls. Scripta Materialia. 46 (5), 379-382 (2002).
- Dairon, J., Gaillard, Y., Tissier, J. C., Balloy, D., Degallaix, G. Parts Containing Open-Celled Metal Foam Manufactured by the Foundry Route: Processes, Performances and Applications. Advanced Engineering Materials. 13 (11), 1066-1071 (2011).
- LeMay, J. D., Hopper, R. W., Hrubesh, L. W., Pekala, R. W. Low-Density Microcellular Materials. Materials Research Society Bulletin. 15 (12), 19-20 (1990).
- Seliger, H., Deuther, U. Die Herstellung von Schaum- und Zellaluminium. Feiburger Forschungshefte. , 103-129 (1965).
- Kuchek, H. A. Method of Making Porous Metallic Article. US patent. , (1966).
- Han, F., Cheng, H., Wang, J., Wang, Q. Effect of pore combination on the mechanical properties of an open cell aluminum foam. Scripta Materialia. 50 (1), 13-17 (2004).
- Cao, X. -. q., Wang, Z. -. h., Ma, H. -. w., Zhao, L. -. m., Yang, G. -. t. Effects of cell size on compressive properties of aluminum foam. Transactions of Nonferrous Metals Society of China. 16, 351-356 (2006).
- Abdulla, T., Yerokhin, A., Goodall, R. Effect of plasma electrolytic oxidation coating on the specific strength of open-cell aluminium foams. Materials & Design. 32, 3742-3749 (2011).
- San Marchi, C., Mortensen, A., Clyne, T. W., Simancik, F. Fabrication and Comprehensive Response of Open-cell Aluminum Foams with Sub-millimeter Pores. Euromat99. 5, 34 (1999).
- San Marchi, C., Mortensen, A. Deformation of open-cell aluminium foam. Acta Materialia. 49 (19), 3959-3969 (2001).
- Goodall, R., Despois, J. F., Mortensen, A. Sintering of NaCl powder: Mechanisms and first stage kinetics. Journal of the European Ceramic Society. 26 (16), 3487-3497 (2006).
- Despois, J. F., Conde, Y., San Marchi, C., Mortensen, A. Tensile Behaviour of Replicated Aluminium Foams. Advanced Engineering Materials. 6 (6), 444-447 (2004).
- Zhao, Y. Y. Stochastic Modelling of Removability of NaCl in Sintering and Dissolution Process to Produce Al Foams. Journal of Porous Materials. 10 (2), 105-111 (2003).

