14.7:
Principio di Le Chatelier: cambiare la concentrazione
14.7:
Principio di Le Chatelier: cambiare la concentrazione
Un sistema in equilibrio è in uno stato di equilibrio dinamico, con reazioni avanti e indietro che si svolgono a tassi uguali. Se un sistema di equilibrio è sottoposto a un cambiamento delle condizioni che influisce in modo diverso su questi tassi di reazione (uno stress), allora i tassi non sono più uguali e il sistema non è in equilibrio. Il sistema sperimenterà successivamente una reazione netta nella direzione di un tasso maggiore (uno spostamento)che ristabilirebbe l’equilibrio. Questo fenomeno è riassunto dal principio di Le Châtelier: se un sistema di equilibrio viene stressato, il sistema sperimenterà uno spostamento in risposta allo stress che ri stabilisce l’equilibrio.
I tassi di reazione sono influenzati principalmente dalle concentrazioni, come descritto dalla legge di velocità della reazione, e dalla temperatura, come descritto dall’equazione di Arrhenius. Di conseguenza, i cambiamenti di concentrazione e temperatura sono le due sollecitazioni che possono spostare un equilibrio.
Effetto di un cambiamento di concentrazione
Se un sistema di equilibrio è sottoposto a un cambiamento nella concentrazione di una specie reagente o di prodotto, la velocità della reazione avanti o inversa cambierà. Ad esempio, si consideri la reazione di equilibrio:

Quando questo sistema è in equilibrio, i tassi di reazione avanti e indietro sono uguali.
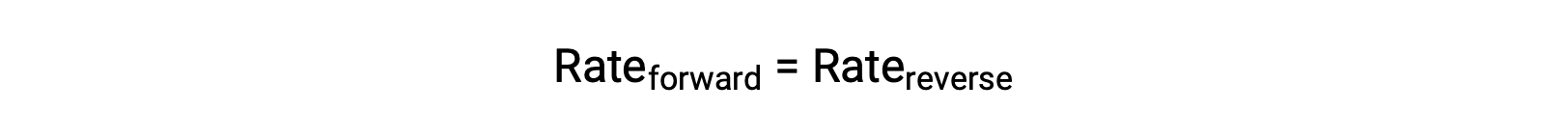
Se il sistema viene stressato aggiungendo reagente, N2 o O2, il conseguente aumento di concentrazione fa aumentare la velocità della reazione in avanti, superando quella della reazione inversa:

Il sistema sperimenterà una reazione netta temporanea nella direzione in avanti per ristabilire l’equilibrio(l’equilibrio si sposterà a destra). Questo stesso cambiamento si verifica se qualche prodotto NO viene rimosso dal sistema, il che diminuisce la velocità della reazione inversa, con conseguente nuovo squilibrio nei tassi.
La stessa logica può essere usata per spiegare lo spostamento a sinistra che deriva dalla rimozione del reagente o dall’aggiunta di prodotto a un sistema di equilibrio. Entrambe queste sollecitazioni si traducono in un aumento del tasso di reazione inversa

e una reazione netta temporanea nella direzione inversa per ristabilire l’equilibrio.
In alternativa a questa interpretazione cinetica, l’effetto dei cambiamenti di concentrazione sugli equilibri può essere razionalizzato in termini di quozienti di reazione. Quando il sistema è in equilibrio,
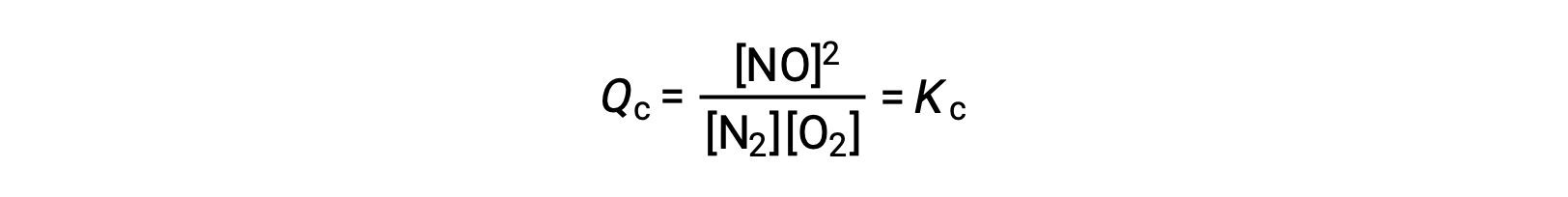
Se viene aggiunto un reagente (aumentando il denominatore del quoziente di reazione) o il prodotto viene rimosso (diminuendo il numeratore), allora Qc < K c el’equilibrio si sposterà a destra. Si noti che i tre diversi modi di indurre questa sollecitazione si traducono in tre diversi cambiamenti nella composizione della miscela di equilibrio. Se si aggiunge N2, lo spostamento destro consumerà O2 e produrrà NO man mano che l’equilibrio viene ro stabilito, producendo una miscela con una concentrazione maggiore di N2 e NO e una concentrazione minore di O2 rispetto a quella presente in precedenza. Se si aggiunge O2, la nuova miscela di equilibrio avrà maggiori concentrazioni di O2 e NO e una concentrazione minore di N2. Infine, se il NO viene rimosso, la nuova miscela di equilibrio avrà concentrazioni maggiori di N2 e O2 e una minore concentrazione di NO. Nonostante queste differenze di composizione, il valore della costante di equilibrio sarà lo stesso dopo lo stress di prima (secondo la legge dell’azione di massa). La stessa logica può essere applicata per le sollecitazioni che coinvolgono la rimozione di reagenti o l’aggiunta di prodotto, nel qual caso Qc > K ce l’equilibrio si sposterà a sinistra.
Questo testo è stato adattato da Openstax, Chemistry 2e, Section 13.3 Shifting Equilibria: LeChatelier’s Principle.
