Modeling Spontaneous Metastatic Renal Cell Carcinoma (mRCC) in Mice Following Nephrectomy
Summary
Models of spontaneous metastatic renal cell carcinoma (RCC) disease progression can be used for evaluating treatments in a clinically relevant setting. This protocol demonstrates different procedures for orthotopic kidney tumor cell implantation, proper nephrectomy, and finally outlines a necropsy guide for visual and bioluminescent scoring of metastatic burden and localization.
Abstract
One of the key challenges to improved testing of new experimental therapeutics in renal cell carcinoma (RCC) is the development of models that faithfully recapitulate early- and late-stage metastatic disease progression. Typical tumor implantation models utilize ectopic or orthotopic primary tumor implantation, but few include systemic spontaneous metastatic disease that mimics the clinical setting. This protocol describes the key steps to develop RCC disease progression stages similar to patients. First, it uses a highly metastatic mouse tumor cell line in a syngeneic model to show orthotopic tumor cell implantation. Methods include superficial and internal implantation into the sub-capsular space with cells combined with matrigel to prevent leakage and early spread. Next it describes the procedures for excision of tumor-bearing kidney (nephrectomy), with critical pre- and post- surgical mouse care. Finally, it outlines the steps necessary to monitor and assess micro-and macro-metastatic disease progression, including bioluminescent imaging as well provides a detailed visual necropsy guide to score systemic disease distribution. The goal of this protocol description is to facilitate the widespread use of clinically relevant metastatic RCC models to improve the predictive value of future therapeutic testing.
Introduction
The major cause of mortality in patients with renal cell carcinoma (RCC) is systemic metastatic disease that typically occurs following surgical removal of a primary tumor growing in the kidney. However, very few preclinical tumor models evaluating experimental therapeutics in mice include metastatic disease, and fewer still faithfully recapitulate the clinical stages of localized growth, surgery, and spontaneous micro-metastatic initiation and progression 1-3. This gap in testing has become increasingly important in the evaluation of new therapies as sometimes striking anti-tumor effects seen in animal models have not always translated into similarly successful treatment of patients 4. Such differences in results may stem from differential drug efficacies between localized ectopic or orthotopic primary tumor models and late-stage metastatic disease 5-7. In the case of RCC, only a few studies have employed established animal protocols that include spontaneous recurrent disease that mimics patients who typically have had tumor-bearing kidneys wholly or partially removed 2,3. The reasons for this dearth in mouse model testing vary. First, there is the high animal cost and inherent variability of tumor cell selection and metastatic potential. For example, human kidney cell lines tend to rarely metastasize, and must be selected over multiple rounds of orthotopic primary implantation and metastatic selection to derive variants that consistently disseminate and form distant lesions (see description of such a human cell line derivation 8-10). Conversely, mouse cells in immunocompetent models tend to behave aggressively, and low cell numbers must be injected with matrigel to reduce immediate systemic spread 3. Second, technical difficulties in performing proper implantation, surgical resection (nephrectomy), and tracking (and quantifying) spontaneous metastatic growth can be challenging and several critical variables need consideration when employing this technique (see Discussion for details). The purpose of this protocol is to describe the essential steps (as well as potential pitfalls) of orthotopic implantation, resection (nephrectomy), and monitoring of spontaneous metastatic RCC disease and to offer a guideline for standardized (and more widespread) use among scientific laboratories that assess the efficacy of experimental therapeutics.
Protocol
1. Orthotopic Kidney Tumor Implantation
- Cell Culture
- Prior to orthotopic implantation, grow mouse RENCALUC cells as a monolayer to 75% confluency.
- Following trypsinization and resuspension in 5% FBS containing media, centrifuge cells at 1,000 rpm, 4 °C for 5 min, repeating 3 times to wash in PBS. Then resuspend the cells in serum-free media to a concentration of 5 x 104 RENCALUC/5 μl serum-free media.
Note: Depending on the variant or cell line used, cells can be resuspended in a 1:1 ratio of matrigel and serum free media to slow highly aggressive mouse cell growth and dissemination after implantation.
- Surgery and cell implantation
Note: All animal studies described therein, including maintenance and determination of experimental endpoints, were performed according to an Institutional Animal Care and Use Committee (IACUC) protocol approved by Roswell Park Cancer Institute.- Anesthetize one Balb/C mouse using Isoflurane (2-3%). Pinch foot and check for reflex to ensure sufficient anesthetization. Apply vet ointment to the eyes to prevent drying. Place mouse in right lateral recumbency and shave left side between fore- and hind-limb. Apply alcohol and iodine along dorsal lumbar area to aseptically prepare area for incision.
- Use surgical scissors to initiate a 1 cm skin incision in a longitudinal direction between the last rib and the hip joint. Loosen connective tissue under the skin using a blunt dissection scissor. Use surgical scissors to make a 0.5 cm incision in a longitudinal direction in the abdominal wall. Use curved forceps to gently push down around open wound – this will exteriorize the kidney and allow for gentle immobilization of organ prior to injection.
- Load the Hamilton syringe with 5 μl the prepared cell mixture. For sub-capsular implantations, two implantation methods can be employed:
- Superficial: Running parallel to the longitudinally orientated kidney, insert needle under the kidney capsule (but above the parenchyma) with the beveled edge up. Once needle is in place, inject all of the cells until small white bubble forms. Slowly remove needle from capsule and immediately blot over injection with a sterile cotton tipped applicator to absorb any leakage and prevent cell dissemination.
- Internal: With beveled edge up and starting from the side of kidney opposite to final implantation site, insert needle through interior of kidney until needle is visible (but not puncturing) the sub-capsular space. Inject all of the cells until white bubble forms and swab injection site to prevent any leakage.
- Gently return the kidney to the body cavity. Close the abdominal body wall using absorbable 5-0 Vicryl suture. Once complete, pull the skin layer together and place 2-3 wound clips. Ensure clips are firm and evenly spaced to ensure no wound exposure. Prior to recovery administer 500 μl of 0.9% NaCl and 100 μl of Buprenorphine (0.01 mg/ml) subcutaneously using a 25 G needle. Routinely monitor clip placement and stability, and remove after 10 days.
2. Nephrectomy/Primary Tumor Removal
- Follow identical protocol for mouse anesthetization, immobilization, incision, and exposure of kidney as described in sections 1.2.1 and 1.2.2 (above).
- Use a second pair of forceps to isolate the kidney by gently removing the connecting fat tissue from the caudal end and the adrenal gland from the cranial end of the kidney. While gently grasping the kidney, use absorbable 5-0 Vicryl suture to make a double knot around the ureter, renal artery, and vein. Slowly secure and then cut above the knot to remove the kidney. If any sign of immediately blood loss, or there is a risk of insufficient hold of suture on ureter, artery, and vein, then use cauterizer instead of scissors to cut above knot.
- Once kidney is removed, carefully check for any bleeding from the tied artery and use cauterizer if necessary. Close the abdominal body wall using 5-0 Vicryl. Pull the skin layer together and place 2-3 wound clips following same precautions as described in 1.2.4. Closely monitor animal daily following surgery for signs of distress, bleeding, or limited mobility. Follow institutional guidelines for animal safely.
3. Monitoring Metastatic Progression and Localization at Endpoint
- Metastatic disease quantification: Bioluminescent monitoring
- See methodology previously described in JOVE for monitoring metastasis using cells transfected with luciferase and quantified using the Xenogen IVIS imaging system 11,12.
- Visual necropsy guide for comparative assessment of metastatic distribution
- Sacrifice animals according to institutional guidelines using endpoints including signs of distress, labored breathing, weight loss, etc.
Note: Close monitoring of animals is critical as spontaneous metastatic progression can be highly variable and rapid. - Use surgical scissors to initiate an incision above the urethra that continues up the thoracic cavity to lower mandible. Use blunt-ended scissors to separate the skin from the muscle of the abdominal wall.
- Initiate incisions along arms and legs. Peel skin away. Closely examine the skin fascia and abdominal wall for nodules, then the superficial lymph nodes: inguinal, brachial, axillary and superficial cervical.
- Make an incision in the abdominal wall and cut along the left and right lateral sides up to the diaphragm. With one horizontal cut remove the front portion of the abdominal wall to bare the viscera.
- Examine the undisturbed appearance of the viscera for grossly notable tumors. Also note the contents of the stomach and intestines; if they are empty, the mouse may not have been able to eat. If the contents are dark, this may indicate evidence of upper/lower gastrointestinal hemorrhage.
- Visually score each organ for tumor presence or absence, assigning a total score for each mouse group (4 animals per group is used as example in Figure 1D-i).
Notes: 1) Enlargement of lymph nodes does not necessarily mean that it is a tumor growth; if it is greatly sized, very firm, and a whitish color, it is likely a metastatic nodule. 2) Excessive milky or cloudy liquid in the abdomen indicates ascites. Ascites may include solid accumulations on the abdominal organs, particularly between the lobes of the liver and the stomach. 3) for organs such as the lungs, multiple nodules can be present and counted for comparisons between individual animals (see 13 for example). - Identify gross tumors or abnormalities of the individual abdominal organs: liver, spleen, kidneys and pancreas by carefully removing and washing with PBS from a squirt bottle.
- After removing the abdominal organs, inspect the abdominal lymph nodes, including the lumbar, sacral, renal, and mesenteric lymph node.
- Assess diaphragm for evidence of metastatic spread.
Note: Extensive spread on the diaphragm can cause it to lose its contractility, leading to labored or loss of breathing. - Remove diaphragm and make two lateral incisions in the thoracic cavity, removing the frontal portion to expose the lungs and heart.
- Remove lungs and heart by gently grasping and lifting the esophagus and cutting the trachea above the heart. Assess tissues for nodules. Squeeze heart. Lack of springy consistency may indicate tumor present.
- Use surgical scissors to cut linearly through skull. Gently remove small pieces of cranium until the whole brain is exposed. Look throughout brain for visible nodules or abnormalities discolorations.
- Sacrifice animals according to institutional guidelines using endpoints including signs of distress, labored breathing, weight loss, etc.
Representative Results
Figure 1A shows a schematic outlining the procedures detailed in this protocol summary. Several important factors must be considered for each step. For example, in Step 1 it shows two methods for sub-capsular tumor cell implantation into the kidney. Tumor cells can be implanted into the sub-capsular space with a small white-bubble confirming the localized placement of cells with leakage prevented by careful removal of needle and swabbing excess escaping fluid (Figure 1B-i). To prevent leakage, cells can be implanted internally first to reach the sub-capsular space (Figure 1B-ii), and combined with matrigel to prevent the initial distribution of tumor cells into the bloodstream. For surgical removal of kidney, exact timing depends on the growth and metastatic potential of the cell line used. Optimal nephrectomy practice includes a fast, but controlled, surgical procedure to minimize mouse distress and recovery. Tight and secure knotting of Vicryl to close ureter, renal artery and vein is key to avoid hemorrhaging (see Figure 1C). Cauterization should be used, but only if potential of excess bleeding requires intervention. Finally, tracking of spontaneous disease by non-invasive bioluminescent assessment allows continued monitoring of occult lesions and growth, as well as quantification at endpoint (see Figure 1D). However, the use of luciferase-labeled cells in spontaneous metastasis models can sometimes lead to clonal tumor cell selection of variants that may not express the transfected gene, and therefore may not always be visible after multiple rounds of selection or long-term tumor growth. In this case, clinically relevant histological analysis can be performed to identify micro-metastatic lesions or, if not technically (of financially) feasible, a visual scoring method to assess visual macro-metastatic disease can be used as a superficial measure of presence/absence of lesions in organ tissue (Figures 1D-i and 1D-ii).
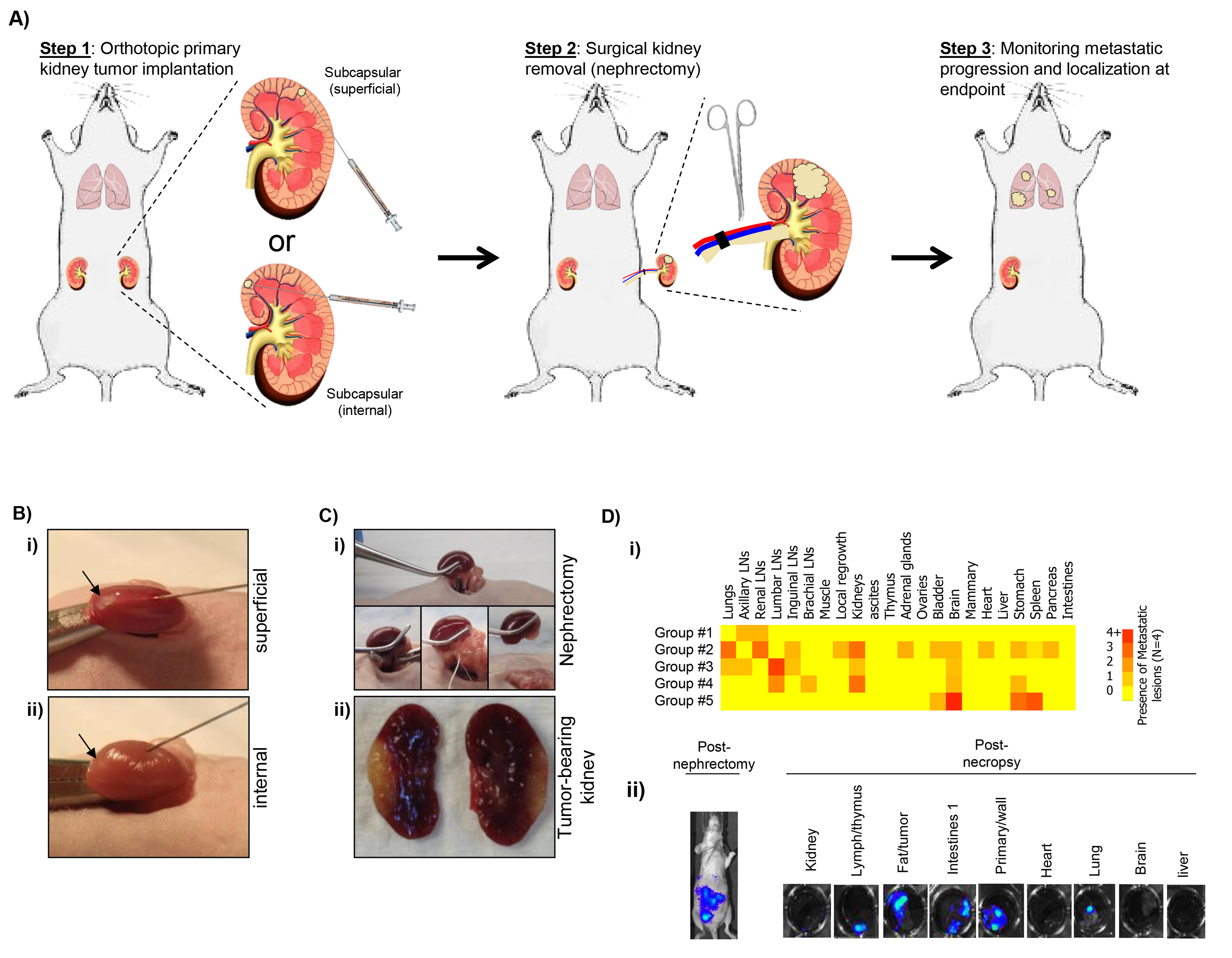
Figure 1. A) Schematic of surgical procedures including implantation (Step 1), surgical resection/nephrectomy (Step 2), and evaluation of post-surgical metastatic disease (Step 3). B) Example of sub-capsular implantation including (i) superficial and (ii) internal technique. C) Example of surgical nephrectomy procedure (i) and representative image of excised kidney with implanted tumor (ii). D) Assessment of post-surgical spontaneous metastatic disease using i) a visual scoring system to assess disease distribution (representative results from 5 groups of 4 animals), and ii) bioluminescent data immediately before and after sacrifice to quantitatively assess micro- and macro-metastatic lesions. Click here to view larger image.
Discussion
The purpose of this protocol is to assess clinically relevant spontaneous metastatic disease using a syngeneic tumor mouse model to describe implantation/resection techniques. Currently, the majority of preclinical studies evaluating novel experimental therapeutics do not include study of metastatic disease and only few recapitulate the stages of primary tumor growth, surgical resection, and eventual spontaneous metastatic spread. To date, genetically engineered mouse models (GEMMs) generated to elicit spontaneous renal cell disease progression (see 14 for summary) do not include late stage metastasis and surgical intervention strategies have thus far not been reported. Therefore, the study of spontaneous metastatic RCC is presently restricted to models of orthotopic tumor cell implantation. Though these models have been previously developed 2,3, this protocol is not widely used in the evaluation of new treatments, particularly in the perioperative setting where it may have the most predictive value. This protocol aims to detail a step-by-step guide of the necessary procedures that may facilitate more widespread use in therapeutic evaluations. In terms of potential pitfalls in performing the techniques as described, there are several key considerations that could influence outcomes. First, tumor cell leakage following implantation could lead to peritoneal disease dissemination and therefore not represent initial localized primary growth. Second, optimal resection times must be established for each cell line used. This is important because surgical nephrectomy of tumor-bearing kidney too early may not allow for disease dissemination (i.e. it may be curative), and surgery too late may lead to non-localized disease and organ-fusing (typically to spleen), making surgery and initial mouse recovery not feasible. Finally, monitoring spontaneous metastatic disease with bioluminescence must be concurrent with strict adherence to institutional guidelines monitoring signs of animal morbidity as disease can be highly variable in terms of both localization and overall impact. Proper statistical powering of animal experiments will allow for all variability to be included and will more faithfully recapitulate clinical disease progression and treatment responses. Taken together, in this article we have demonstrated techniques used to successfully recapitulate localized orthotopic RCC and post-surgical disease recurrence with the aim of improving clinical relevance in preclinical tumor models
Disclosures
The authors have nothing to disclose.
Acknowledgements
We are grateful to the laboratory of Dr. Robert S. Kerbel (University of Toronto, Sunnybrook Research Institute, Toronto, Canada) for technical help and expertise in the development of this procedure. We would also like to thank Dr. Sandra Sexton and the Roswell Park Cancer Institute Department of Laboratory Animal Resources. This work was supported by an award from the Roswell Park Alliance Foundation (to JMLE).
Materials
| DMEM-high glucose with Pyr. And L-Glutamine | Corning | 10-013-CV | |
| FBS | Invitrogen | 10437-028 | |
| 0.25% Trypsin EDTA | Corning | 25-053-CL | |
| 1x DPBS without Calcium & Magnesium | Corning | 21-031-CV | |
| Matrigel | BD Biosciences | 354234 | must be kept on ice |
| Artifical tears-lubricant opthalmic ointment | Akorn Animal Health | 17478-162-35 | |
| Pocket pro pet trimmer | Braintree scientific | CLP9931B | |
| Alcohol swab | VWR | 326895 | |
| Betadine solution swab | VWR | 67618-152-01 | |
| MICRO DISSECTING sissors straight,blunt – 25mm blades – 4.5" | Southpointe surgical | RS-5982 | |
| Iris Forceps, serrated, curved, 10cm long | Kent scientific | INS15915 | need two of these |
| 10µl Hamilton syringe | Hamilton | 7635-01 | |
| 30G, 45 degree, RN needle | Hamilton | 7803-07 | |
| Sterile cotton tipped appicator | VWR | 10805-144 | |
| High temperature cautery kit | Kent scientific | INS500392 | |
| 5-0 coated Vicryl, conventional cutting needle | Ethicon | J834 | |
| Reflex clip applier for 7mm clips | Kent scientific | INS500343 | |
| Reflex clips, 7mm, non-sterile | Kent scientific | INS500344 | |
| Removing forceps, 12cm lone | Kent scientific | INS500347 | |
| 0.9% Sodium Chloride | Baxter Healthcare | 2B1322 | |
| Buprenorphine 0.01mg/mL | |||
| 25G 5/8" needle | VWR | BD305122 | |
| 1mL syringe w/out needle | VWR | BD309659 | |
| D-Luciferin | Gold Bio technology | LUCK-1G |
References
- Francia, G., Cruz-Munoz, W., Man, S., Xu, P., Kerbel, R. S. Mouse models of advanced spontaneous metastasis for experimental therapeutics. Nat. Rev. Cancer. 11, 135-141 (2011).
- Souza, B. M., Chaves, K. B., Chammas, R., Schor, N., Bellini, M. H. Endostatin neoadjuvant gene therapy extends survival in an orthotopic metastatic mouse model of renal cell carcinoma. Biomed. Pharmacother. 66, 237-241 (2012).
- Amagai, Y., et al. Combination therapy of interleukin-2 and sorafenib improves survival benefits and prevents spontaneous pulmonary metastasis in murine renal cell carcinoma models. Jpn. J. Clin. Oncol. 40, 503-507 (2010).
- Steeg, P. S., et al. Preclinical Drug Development Must Consider the Impact on Metastasis. Clin. Cancer Res. 15, 4529-4530 (2009).
- Guerin, E., Man, S., Xu, P. A model of postsurgical advanced metastatic breast cancer more accurately replicates the clinical efficacy of antiangiogenic drugs. Cancer Res. 73, 2743-2748 (2013).
- Day, C. P., Carter, J., Bonomi, C., Hollingshead, M., Merlino, G. Preclinical therapeutic response of residual metastatic disease is distinct from its primary tumor of origin. Int. J. Cancer. 130, 190-199 (2012).
- Ebos, J. M., Kerbel, R. S. Antiangiogenic therapy: impact on invasion, disease progression, and metastasis. Nat. Rev. Clin. Oncol. 8, 210-221 (2011).
- Naito, S., Walker, S. M., Fidler, I. J. In vivo selection of human renal cell carcinoma cells with high metastatic potential in nude mice. Clin. Exp. Metastasis. 7, 381-389 (1989).
- Fidler, I. J., Naito, S., Pathak, S. Orthotopic implantation in essential for the selection, growth and metastasis of human renal cell cancer in nude mice. Cancer Metastasis Rev. 9, 145-165 (1990).
- Saiki, I., et al. Characterization of the invasive and metastatic phenotype in human renal cell carcinoma. Clin. Exp. Metastasis. 9, 551-566 (1991).
- Mohanty, S., Xu, L. Experimental metastasis assay. J. Vis. Exp. 42 (1942), (2010).
- Nunez-Cruz, S., Connolly, D. C., Scholler, N. An orthotopic model of serous ovarian cancer in immunocompetent mice for in vivo tumor imaging and monitoring of tumor immune responses. J. Vis. Exp. (45), (2010).
- Ebos, J. M., et al. Accelerated metastasis after short-term treatment with a potent inhibitor of tumor angiogenesis. Cancer Cell. 15, 232-239 (2009).
- Yang, O. C., Maxwell, P. H., Pollard, P. J. Renal cell carcinoma: translational aspects of metabolism and therapeutic consequences. Kidney Int. 84 (4), 667-681 (2013).

