Ultraviolet-Visible (UV-Vis) Spectroscopy
English
Share
Overview
Source: Laboratory of Dr. B. Jill Venton – University of Virginia
Ultraviolet-visible (UV-Vis) spectroscopy is one of the most popular analytical techniques because it is very versatile and able to detect nearly every molecule. With UV-Vis spectroscopy, the UV-Vis light is passed through a sample and the transmittance of light by a sample is measured. From the transmittance (T), the absorbance can be calculated as A=-log (T). An absorbance spectrum is obtained that shows the absorbance of a compound at different wavelengths. The amount of absorbance at any wavelength is due to the chemical structure of the molecule.
UV-Vis can be used in a qualitative manner, to identify functional groups or confirm the identity of a compound by matching the absorbance spectrum. It can also be used in a quantitative manner, as concentration of the analyte is related to the absorbance using Beer's Law. UV-Vis spectroscopy is used to quantify the amount of DNA or protein in a sample, for water analysis, and as a detector for many types of chromatography. Kinetics of chemical reactions are also measured with UV-Vis spectroscopy by taking repeated UV-Vis measurements over time. UV-Vis measurements are generally taken with a spectrophotometer. UV-Vis is also a very popular detector for other analytical techniques, such as chromatography, because it can detect many compounds.
Typically, UV-Vis is not the most sensitive spectroscopy technique, because not a lot of light is absorbed over a short path length. Other spectroscopy techniques such as fluorescence have higher sensitivity, but they are not as generally applicable, as most molecules are not fluorescent. UV-Vis has a similar sensitivity to other absorbance measurements, such as infrared spectroscopy.
Principles
UV-Vis is often called a general technique because most molecules will absorb in the UV-Vis wavelength range. The UV extends from 100–400 nm and the visible spectrum from 400–700 nm. The 100–200 nm range is called the deep UV. Light sources are more difficult to find for this range, so it is not routinely used for UV-Vis measurements. Typical UV-Vis spectrometers use a deuterium lamp for the UV that produces light from 170–375 nm and a tungsten filament lamp for visible, which produces light from 350–2,500 nm.
When a photon hits a molecule and is absorbed, the molecule is promoted into a more excited energetic state. UV-visible light has enough energy to promote electrons to a higher electronic state, from the highest occupied molecular orbital (HOMO) to the lowest unoccupied molecular orbital (LUMO). The energy difference between the HOMO and the LUMO is called the band gap. Typically, these orbitals are called bonding and anti-bonding. The energy of the photon must exactly match the band gap for the photon to be absorbed. Thus, molecules with different chemical structures have different energy band gaps and different absorption spectra. The most common transitions that fall in the UV-Vis range are π-π* and n- π*. Pi orbitals arise due to double bonds, and n orbitals are for non-bonding electrons. Pi star are anti-bonding pi orbitals. Thus, the best UV-Vis absorption is by molecules that contain double bonds. Pi orbitals adjacent to each other that are connected, called conjugation, typically increases absorption. Sigma-σ* transitions, associated with single bonds, are higher energy and fall in the deep UV, so they are less useful for routine use. The appearance of broad bands or shoulders on the UV-Vis structure is due to the numerous vibrational and rotational states of a molecule, which lead to separate energy band gaps of slightly different energies.
For molecules with absorption in the visible region, the compounds will often appear colored. However, a common misconception is that the wavelength of peak absorption (λmax) for a compound is the color it appears. A compound that appears red does not have much absorption in the red region of the spectrum. Instead, the λmax for a compound that looks red is green. The color of a compound arises because those wavelengths of light are selectively transmitted through the sample, and thus they are not absorbed. A color wheel is helpful in determining what color a compound will absorb and what range the λmax will be, as the color directly across the wheel from the observed color is the color that is most absorbed.
Absorption follows Beer's Law, A=εbC where ε is the molar attenuation coefficient, b is path length, and C is concentration. The molar attenuation coefficient is the characteristic of an individual compound to absorb at a given wavelength and this property is due to functional groups, conjugation, etc. If a compound does not have a high attenuation coefficient, it could be tagged with an appropriate group to increase its absorbance. Path length is generally related to the size of the cuvette and is 1 cm in standard spectrophotometers.
UV-Vis is performed on a variety of instruments, from traditional spectrophotometers to more modern-day plate readers. The absorbance wavelength must be chosen, either using a filter or a monochromator. A monochromator is a device that separates the wavelengths of light spatially and then places an exit slit where the desired wavelength of light is. Monochromators can be scanned to provide a whole absorbance spectrum. Alternatively, a diode-array instrument allows all colors of light to be transmitted through the sample, then the light is separated into different wavelengths spatially and detected using photodiodes. Diode-array instruments collect full spectra faster, but are more complicated and more expensive.
Procedure
1. Calibrate the Spectrometer
- Turn on the UV-Vis spectrometer and allow the lamps to warm up for an appropriate period of time (around 20 min) to stabilize them.
- Fill a cuvette with the solvent for the sample and make sure the outside is clean. This will serve as a blank and help account for light losses due to scattering or absorption by the solvent.
- Place the cuvette in the spectrometer. Make sure to align the cuvette properly, as often the cuvette has two sides, which are meant for handling (may be grooved) and are not meant to shine light through.
- Take a reading for the blank. The absorbance should be minimal, but any absorbance should be subtracted out from future samples. Some instruments might store the blank data and perform the subtraction automatically.
2. Perform an Absorbance Spectrum
- Fill the cuvette with the sample. To make sure the transfer is quantitative, rinse the cuvette twice with the sample and then fill it about ¾ full. Make sure the outside is clean of any fingerprints, etc.
- Place the cuvette in the spectrometer in the correct direction.
- Cover the cuvette to prevent any ambient light.
- Collect an absorbance spectrum by allowing the instrument to scan through different wavelengths and collect the absorbance. The wavelength range can be set with information about the specific sample, but a range of 200–800 nm is standard. A diode-array instrument is able to collect an entire absorbance spectrum in one run.
- From the collected absorbance spectrum, determine the absorbance maximum (λmax). Repeat the collection of spectra to get an estimate of error in λmax.
- To make a calibration curve, collect the UV-Vis spectrum of a variety of different concentration samples. Spectrometers are often limited in linear range and will not be able to measure an absorbance value greater than 1.5. If the absorbance values for the sample are outside the instrument's linear range, dilute the sample to get the values within the linear range.
3. Kinetics Experiments with UV-Vis Spectroscopy
- UV-Vis can be used for kinetics experiments by examining the change in absorbance over time. For a kinetics experiment, take an initial reading of the sample.
- Quickly add the reagent to start the chemical reaction.
- Stir it well to mix with the sample. If a small amount is added, this could be done in a cuvette. Alternatively, mix the reagent with sample and quickly pour some in a cuvette for a measurement.
- Measure the absorbance at the λmax for the analyte of interest over time. If using up the reagent being measuring (i.e. absorbance is going up because there is less reagent to absorb), then the decay will indicate the order of the reaction.
- Using a calibration curve, make a plot of analyte concentration vs time, converting the absorbance value into concentration. From there, this graph can be fit with appropriate equations to determine the reaction rate constants.
Ultraviolet-visible, or UV-Vis, spectroscopy is one of the most popular analytical techniques in the laboratory.
In UV-Vis spectroscopy, light is passed through a sample at a specific wavelength in the UV or visible spectrum. If the sample absorbs some of the light, not all of the light will be pass through, or be transmitted. Transmission is the ratio of the intensity of the transmitted light to the incident light, and is correlated to absorbance. The absorbance can be used in a quantitative manner, to obtain the concentration of a sample. It can also be used in a qualitative manner, to identify a compound by matching the measured absorbance over a range of wavelengths, called the absorbance spectrum, to the published data. This video will introduce UV-Vis spectroscopy, and demonstrate its use in the laboratory in determining sample concentration and reaction kinetics.
When a photon hits a molecule and is absorbed, the molecule is promoted from its ground state into a higher energy state. The energy difference between the two is the band gap. The energy of the photon must exactly match the band gap in order for the photon to be absorbed. The chemical structure determines the band gap; therefore molecules each have unique absorbance spectra.
Absorbance follows Beer's Law, which states absorbance equals the molar attenuation coefficient times the path length and concentration. The molar attenuation coefficient is related to the individual compound's ability to absorb light of a specific wavelength. Path length refers to the distance traveled by light through the sample, which is typically 1 cm for standard cuvettes. Beer's law can be used to calculate sample concentration, if the absorptivity is known, or a calibration curve can be used.
UV-Vis is often called a general technique, as most molecules absorb light in the UV-visible wavelength range. The UV range extends from 100–400 nm, and the visible spectrum ranges from 400–700 nm. However, most spectrophotometers do not operate in the deep UV range of 100–200 nm, as light sources in this range are expensive. Most UV-Vis spectrophotometers use a deuterium lamp for the UV range, which produces light from 170–375 nm, and a tungsten filament lamp for the visible range, which produces light from 350–2,500 nm.
Since the light source is usually a lamp with broad wavelength ranges, the specific absorbance wavelength is selected using filters or a monochromator. A monochromator is a device that separates the wavelengths of light spatially, and then places an exit slit where the desired wavelength of light is. The monochromator can be scanned over a wavelength range to provide an entire absorbance spectrum. This makes the technique useful for quantifying and identifying a wide range of molecules.
Now that the basics of UV-Vis spectroscopy have been outlined, lets take a look at a simple UV-Vis experiment in the laboratory.
Before beginning the measurement, turn on the spectrophotometer, and allow the lamps to warm up for an appropriate period of time to stabilize them.
Prepare a blank by filling a clean cuvette with the sample solvent, and then wipe the outside with lint-free paper to remove any fingerprints.
Ensure that the cuvette is aligned properly with any grooved sides out of the beam-path, and insert it into the spectrophotometer. Secure the lid to prevent ambient light from entering the system.
Measure the absorbance of the blank at one wavelength, or over a wavelength range. Record or save the absorbance, as it must be subtracted from the absorbance of the sample.
Next, discard the blank and rinse the cuvette twice with sample. Then, fill the cuvette about ¾ full with sample. Wipe the outside of the cuvette again, to ensure that it is clean and free of fingerprints.
Place the cuvette in the spectrophotometer in the correct orientation, and secure the lid.
Collect an absorbance measurement or spectrum at the same wavelength or wavelength range as the blank. Subtract the blank spectrum or measurement, if the instrument does not automatically do so.
From the collected absorbance spectrum, determine the absorbance maximum, or λmax.
To quantify the amount of analyte in the sample, create a calibration curve using a range of known analyte concentrations. For more information on how to construct and use a calibration curve, please watch this collection's video "Calibration Curves".
The absorbance measurement can also be used to calculate reaction kinetics by measuring the increase or decrease in a compounds concentration throughout the reaction. Begin by taking an initial reading of the sample, blue dye in this case, at the absorbance maximum before the reaction.
Next, quickly add the reagent, bleach in this case, to start the chemical reaction. Stir it well, so that it mixes with the sample.
Measure the absorbance at the absorbance maximum over time.
The initial absorbance spectrum of the blue dye sample is shown. The background colors show the colors of light in the visible spectrum. The blue dye has an absorbance maximum at about 630 nm.
The kinetics of the reaction between blue dye and bleach was measured over time. The absorbance of blue dye decreases over time, as it reacts with the bleach. The absorbance reaches near zero after 300 s, indicating that the reaction has neared completion. For more information on kinetics and reactions, please watch the JoVE Science Education video "Reaction Rate Laws".
UV-Vis spectroscopy is used heavily in many different research areas to identify or quantify a sample.
For example, UV-Vis spectroscopy is used heavily in biological fields to quantify the amount of protein in a sample. A Bradford assay is often used to quantify proteins, with the aid of a dye. First, a calibration curve of known protein concentrations is prepared, typically using Bovine Serum Albumin, or BSA. Then Coomassie blue stain is added to each of the standards and to the sample. The absorbance of the protein-dye complex is then measured at 595 nm.
Alternatively, proteins can be measured directly by their absorbance at 280 nm. In this example, protein concentration is quantified using an ultra low volume spectrophotometer. For many proteins, an absorbance of 1 correlates to a concentration of 1 mg/mL.
UV-Vis spectroscopy is also used to quantify the amount of bacterial cells in a cell culture. For this measurement, the absorbance, or optical density, is measured at 600 nm. Typically, an OD600 measurement of 1 indicates the presence of 8 x 108 bacterial cells per mL. Measuring the cell density throughout culture growth enables the determination of the bacterial growth curve, and can help to identify when a culture is in its exponential growth phase.
Nitrogen oxide and nitrogen dioxide, or NOx, is a by-product of automobile exhaust, and can be harmful to the environment because it forms damaging tropospheric ozone. NOx can be measured by reacting it with a solution of sulfanilic acid and napthyl-ethylenediamine. The resulting solution is a pink colored azo dye molecule, the intensity of which is directly correlated to NOx concentration. This concentration can then be determined using a UV-Vis spectrophotometer.
You've just watched JoVE's introduction to UV-visible spectroscopy. You should now understand the basics of UV-Vis operation, how to measure a sample using a UV-Vis and how to correlate absorbance to sample concentration.
Thanks for watching!
Results
UV-Vis can be used to obtain a spectrum of colored compounds. In Figure 1A, the absorbance spectrum of a blue dye is shown. The background shows the colors of light in the visible spectrum. The blue dye has a λmax absorbance in the orange/red. Figure 1B shows a spectrum of a red dye, with λmax in the green.
Kinetics can be measured from a plot of absorbance at one wavelength over time. Figure 2 shows a plot of the absorbance of a blue dye (at 630 nm) as it reacts with bleach.
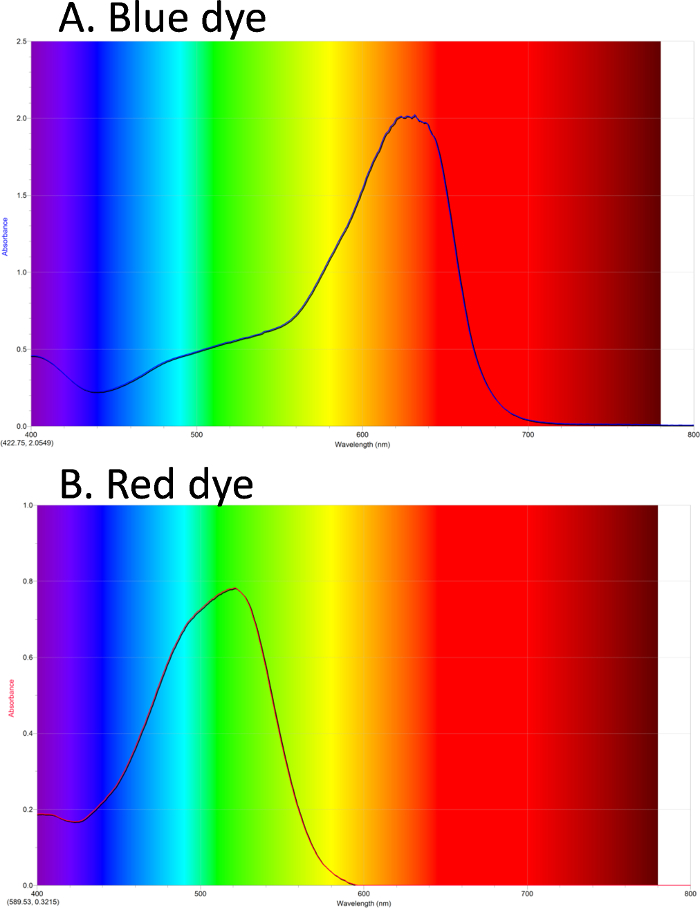
Figure 1. UV-Vis absorbance spectra. A. Blue dye #1 has maximum absorbance in the orange/red. B. Red dye #40 has maximum absorbance in the green. Please click here to view a larger version of this figure.

Figure 2. UV-Vis for kinetics. Absorbance of blue dye #1 as it reacts with bleach. The curve can be fit with an exponential decay, indicating first order kinetics. Please click here to view a larger version of this figure.
Applications and Summary
UV-Vis is used in many chemical analyses. It is used to quantitate the amount of protein in a solution, as most proteins absorb strongly at 280 nm. Figure 3 shows an example spectra of cytochrome C, which has a high absorbance at 280 and also at 450 because of a heme group. UV-Vis is also used as a standard technique to quantify the amount of DNA in a sample, as all the bases absorb strongly at 260 nm. RNA and proteins also absorb at 260 nm, so absorbance at other wavelengths can be measured to check for interferences. Specifically, proteins absorb strongly at 280 nm, so the ratio of absorbance at 280/260 can give a measure of the ratio of protein to DNA in a sample.
Most simple analyses measure the absorbance one wavelength at a time. However, more chemical information is present if measurements are made at many wavelengths simultaneously. Diode-array instruments capture all the light that is transmitted, split the light into different colors using a prism or holographic grating, and then absorbance at different wavelengths is captured on a linear array of photodiodes. The advantage of this method is that it is useful for measuring many different molecules simultaneously.

Figure 3. UV-Vis spectrum of a protein. The peak at 280 nm is indicative of a protein. The peak at 450 is due to absorbance of the heme group in cytochrome C.
Transcript
Ultraviolet-visible, or UV-Vis, spectroscopy is one of the most popular analytical techniques in the laboratory.
In UV-Vis spectroscopy, light is passed through a sample at a specific wavelength in the UV or visible spectrum. If the sample absorbs some of the light, not all of the light will be pass through, or be transmitted. Transmission is the ratio of the intensity of the transmitted light to the incident light, and is correlated to absorbance. The absorbance can be used in a quantitative manner, to obtain the concentration of a sample. It can also be used in a qualitative manner, to identify a compound by matching the measured absorbance over a range of wavelengths, called the absorbance spectrum, to the published data. This video will introduce UV-Vis spectroscopy, and demonstrate its use in the laboratory in determining sample concentration and reaction kinetics.
When a photon hits a molecule and is absorbed, the molecule is promoted from its ground state into a higher energy state. The energy difference between the two is the band gap. The energy of the photon must exactly match the band gap in order for the photon to be absorbed. The chemical structure determines the band gap; therefore molecules each have unique absorbance spectra.
Absorbance follows Beer’s Law, which states absorbance equals the molar attenuation coefficient times the path length and concentration. The molar attenuation coefficient is related to the individual compound’s ability to absorb light of a specific wavelength. Path length refers to the distance traveled by light through the sample, which is typically 1 cm for standard cuvettes. Beer’s law can be used to calculate sample concentration, if the absorptivity is known, or a calibration curve can be used.
UV-Vis is often called a general technique, as most molecules absorb light in the UV-visible wavelength range. The UV range extends from 100–400 nm, and the visible spectrum ranges from 400–700 nm. However, most spectrophotometers do not operate in the deep UV range of 100–200 nm, as light sources in this range are expensive. Most UV-Vis spectrophotometers use a deuterium lamp for the UV range, which produces light from 170–375 nm, and a tungsten filament lamp for the visible range, which produces light from 350–2,500 nm.
Since the light source is usually a lamp with broad wavelength ranges, the specific absorbance wavelength is selected using filters or a monochromator. A monochromator is a device that separates the wavelengths of light spatially, and then places an exit slit where the desired wavelength of light is. The monochromator can be scanned over a wavelength range to provide an entire absorbance spectrum. This makes the technique useful for quantifying and identifying a wide range of molecules.
Now that the basics of UV-Vis spectroscopy have been outlined, lets take a look at a simple UV-Vis experiment in the laboratory.
Before beginning the measurement, turn on the spectrophotometer, and allow the lamps to warm up for an appropriate period of time to stabilize them.
Prepare a blank by filling a clean cuvette with the sample solvent, and then wipe the outside with lint-free paper to remove any fingerprints.
Ensure that the cuvette is aligned properly with any grooved sides out of the beam-path, and insert it into the spectrophotometer. Secure the lid to prevent ambient light from entering the system.
Measure the absorbance of the blank at one wavelength, or over a wavelength range. Record or save the absorbance, as it must be subtracted from the absorbance of the sample.
Next, discard the blank and rinse the cuvette twice with sample. Then, fill the cuvette about ¾ full with sample. Wipe the outside of the cuvette again, to ensure that it is clean and free of fingerprints.
Place the cuvette in the spectrophotometer in the correct orientation, and secure the lid.
Collect an absorbance measurement or spectrum at the same wavelength or wavelength range as the blank. Subtract the blank spectrum or measurement, if the instrument does not automatically do so.
From the collected absorbance spectrum, determine the absorbance maximum, or λmax.
To quantify the amount of analyte in the sample, create a calibration curve using a range of known analyte concentrations. For more information on how to construct and use a calibration curve, please watch this collection’s video “Calibration Curves”.
The absorbance measurement can also be used to calculate reaction kinetics by measuring the increase or decrease in a compounds concentration throughout the reaction. Begin by taking an initial reading of the sample, blue dye in this case, at the absorbance maximum before the reaction.
Next, quickly add the reagent, bleach in this case, to start the chemical reaction. Stir it well, so that it mixes with the sample.
Measure the absorbance at the absorbance maximum over time.
The initial absorbance spectrum of the blue dye sample is shown. The background colors show the colors of light in the visible spectrum. The blue dye has an absorbance maximum at about 630 nm.
The kinetics of the reaction between blue dye and bleach was measured over time. The absorbance of blue dye decreases over time, as it reacts with the bleach. The absorbance reaches near zero after 300 s, indicating that the reaction has neared completion. For more information on kinetics and reactions, please watch the JoVE Science Education video “Reaction Rate Laws”.
UV-Vis spectroscopy is used heavily in many different research areas to identify or quantify a sample.
For example, UV-Vis spectroscopy is used heavily in biological fields to quantify the amount of protein in a sample. A Bradford assay is often used to quantify proteins, with the aid of a dye. First, a calibration curve of known protein concentrations is prepared, typically using Bovine Serum Albumin, or BSA. Then Coomassie blue stain is added to each of the standards and to the sample. The absorbance of the protein-dye complex is then measured at 595 nm.
Alternatively, proteins can be measured directly by their absorbance at 280 nm. In this example, protein concentration is quantified using an ultra low volume spectrophotometer. For many proteins, an absorbance of 1 correlates to a concentration of 1 mg/mL.
UV-Vis spectroscopy is also used to quantify the amount of bacterial cells in a cell culture. For this measurement, the absorbance, or optical density, is measured at 600 nm. Typically, an OD600 measurement of 1 indicates the presence of 8 x 108 bacterial cells per mL. Measuring the cell density throughout culture growth enables the determination of the bacterial growth curve, and can help to identify when a culture is in its exponential growth phase.
Nitrogen oxide and nitrogen dioxide, or NOx, is a by-product of automobile exhaust, and can be harmful to the environment because it forms damaging tropospheric ozone. NOx can be measured by reacting it with a solution of sulfanilic acid and napthyl-ethylenediamine. The resulting solution is a pink colored azo dye molecule, the intensity of which is directly correlated to NOx concentration. This concentration can then be determined using a UV-Vis spectrophotometer.
You’ve just watched JoVE’s introduction to UV-visible spectroscopy. You should now understand the basics of UV-Vis operation, how to measure a sample using a UV-Vis and how to correlate absorbance to sample concentration.
Thanks for watching!
