The Rabbit Blood-shunt Model for the Study of Acute and Late Sequelae of Subarachnoid Hemorrhage: Technical Aspects
Summary
The experimental intracranial pressure-controlled blood shunt subarachnoid hemorrhage (SAH) model in the rabbit combines the standard procedures — subclavian artery cannulation and transcutaneous cisterna magna puncture, which enables close mimicking of human pathophysiological conditions after SAH. We present step-by-step instructions and discuss key surgical points for successful experimental SAH creation.
Abstract
Early brain injury and delayed cerebral vasospasm both contribute to unfavorable outcomes after subarachnoid hemorrhage (SAH). Reproducible and controllable animal models that simulate both conditions are presently uncommon. Therefore, new models are needed in order to mimic human pathophysiological conditions resulting from SAH.
This report describes the technical nuances of a rabbit blood-shunt SAH model that enables control of intracerebral pressure (ICP). An extracorporeal shunt is placed between the arterial system and the subarachnoid space, which enables examiner-independent SAH in a closed cranium. Step-by-step procedural instructions and necessary equipment are described, as well as technical considerations to produce the model with minimal mortality and morbidity. Important details required for successful surgical creation of this robust, simple and consistent ICP-controlled SAH rabbit model are described.
Introduction
Aneurysmal subarachnoid hemorrhage (SAH) is one of the most life threatening neuropathological conditions, frequently leading to permanent neurological damage or death1. Past research has focused on delayed cerebral vasospasm (DCVS) as the primary etiology of neurological deficits associated with SAH2. However, the generally poor clinical outcomes of patients suffering from SAH after treatment of vasospasm has led to an expansion of the research focus to include the effects of early brain injury (EBI) after SAH3. Greater understanding of the significance of both EBI and DCVS in contributing to poor clinical outcomes after SAH is essential for the development of more effective therapeutic strategies.
Up to now, single and double autologous blood injection into the cisterna magna has been the standard method for SAH induction for the study of DCVS2-6. Though commonly used in previous studies, this model most likely does not reproduce the neuropathological key changes associated with SAH induced EBI7. In contrast, endovascular perforation is known to produce severe acute pathophysiological changes that partially mimic the symptoms of EBI7.
This report describes a novel rabbit model of SAH designed to enable investigation of both EBI and DCVS, thereby allowing more accurate characterization of SAH-induced pathology8-10. With the described technique, the standard cisterna magna model is adapted by connecting the arterial system of the subclavian artery and the cisterna magna via an extracorporeal shunt. The blood flow is thereby linked to the rabbit’s physiology and driven by the pressure gradient between the arterial blood and intracranial pressure. The bleeding stops when intracerebral pressure (ICP) equals diastolic blood pressure and the blood in the shunt system coagulates. Utilizing the host’s physiology reduces examiner-dependent SAH induction, leading to a more consistent model of SAH that reliably produces both EBI and DCVS phenotypes3,8-10.
Protocol
Three month old female New Zealand rabbits weighing 2.5 – 3.5 kg were used for this procedure. The study was performed in accordance with the National Institutes of Health guidelines for the care and use of experimental animals and with the approval of the Animal Care Committee of the Canton of Bern, Switzerland (approval #105/13). All surgical procedures were performed under sterile conditions at the Experimental Surgical Institute of the Department of Clinical Research at Bern University Hospital in Bern, Switzerland. A veterinary anesthesiologist monitored the animals during surgery and throughout recovery.
1. Animal Preparation, Positioning and Subclavian Artery Cannulation
- Induce general anesthesia in the rabbit with intramuscular injection of ketamine (30 mg/kg; Ketalar, 50 mg/ml) and xylazine (6 mg/kg; Xylapan 20 mg/ml) and control depth of anesthesia by checking the rabbit’s response to noxious stimulation (e.g., toe pinch). See 1.7 in case of positive response.
- Pull down the lower lids of both eyes and apply a small amount of ointment to the eyelids to prevent dryness and further irritation.
- Catheterize the lateral ear vein with a 20 Gbutterfly venflon (20 G vascular catheter), fix with adhesive tape, and connect to a gravity bag containing 0.9% sodium chloride (500 ml/24 hr) and ketamine (40 mg/kg/hr) / xylazine (4 mg/kg/hr) for continuous intravenous (i.v.) anesthesia. Administer additional analgesics every 15 min i.v. (Fentanyl, 1 mcg/kg). Note: Avoid volatile gas anesthetics, which is associated with decreased CPP, increasing CBF, and decreasing cerebral metabolic rate for oxygen.11 Intravenous anesthetics provide more ideal characteristics for neuroanesthesia by preserving CBF and cerebral vasoconstriction,12 which is of uppermost importance when studying cerebral vasospasm. In addition, although mortality is increased in spontaneously breathing animals, it may better mimic the human situation of acute SAH.
- Provide oxygen (1 – 2 L/min) via a respiratory mask that allows end-tidal carbon dioxide (EtCO2) monitoring.
- Install a 3 channel electrocardiogram (ECG).Place three subcutaneous electrodes in a triangular arrangement on the ventral side of the rabbit; specifically, place one electrode over the right midthoracic region (with distance to the sterile shaved field for the subclavian artery cannulation) and two electrodes in the lower abdomen distributed over both limbs.
- Monitor the depth of anesthesia every 15 min during surgery by following respiratory rate, heart rate (HR) monitored from the ECG signal, and reaction to noxious stimulation.
- In case of positive response to noxious stimulation (toe pinch), adapt the depth of anesthesia by ketamine bolus (6 mg/kg) i.v. and xylazine bolus (0.05 mg/kg) i.v. and/or an additional analgesia bolus with Fentanyl (1 mcg/kg) i.v.
- Fix the rabbit in a supine position on a body warming plate, tilting the head 20° down and turning it slightly contralaterally to the side on which the subclavian artery will be exposed.
- apply eye ointment and prepare the area for surgery by shaving the hair over the right pectoral muscle around the middle third of the clavicle, and over the frontal-, parietal- and occipital skull, the neck, and over the right common femoral artery.
- Disinfect the skin for 3 min with a broad spectrum antiseptic, e.g., Povidone-iodine.
- Cover the rabbit with sterile sheets. Perform all further procedures under sterile conditions and frequently apply 4% papaverin HCl and antibiotic solution (neomycin sulfate 5 mg/ml) topically to prevent arterial vasospasm by vessel manipulation and local infections.
- Infiltrate the pectoral muscle with local anesthetics (Lidocaine 1% maximal 6 mg/kg). Make a parasternal skin incision and prepare the pectoral muscle. Using the microscope, dissect the subclavian artery and secure with a proximal and distal ligature (4-0 polyfilament sutures) around the exposed end. Keep one ligature close to the proximal control in place to secure the catheter and ligate the vessel distally.
- Perform an arteriotomy in the wall of the subclavian artery by incising the artery with a curved microscissor and cannulate the subclavian artery retrogradely with a small intravasal 3-way stopcock. Secure the catheter by double knot ligature towards the distal ligature in order to prevent arterial twisting or bending of the proximal part of the artery and to avoid slippage or massive bleeding.
2. Blood Pressure and Arterial Blood Gas Monitoring
- Connect the 3-way stop cock to i) the intravasal cannula for arterial blood gas (ABG) analyses, pH, PaCO2, PaO2, bicarbonate, base excess, and SO2, ii) the invasive arterial blood pressure measurement device, and iii) the shunt device.
- Collect blood samples for ABG status (PaCO2, PaO2) and continuously monitor standard cardiovascular and respiratory parameters (blood pressure, HR, ECG, respiratory rate and end-tidal CO2) and transfer data via the analog output interface to an analog–digital converter/data logger and store.
NOTE: The pressures will be zeroed at heart levels before and after each session, and pressure calibration of the analog/digital-converter and data-logging system will be done once before the series starts.
3. Baseline Digital Subtraction Angiography
- Place an external sizing device (small sphere) over both mandible angles in order to calibrate the angiogram.
NOTE: This allows exact comparison of post hoc measurements of baseline and follow up vessel diameter. - Perform digital subtraction angiography (DSA) by retrograde intra-arterial bolus injection of non-ionic Iopamidol (0.6 ml/kg, 5 ml/sec for 2 sec) through the cannulated artery and flush the cannula immediately after bolus injection with saline in order to prevent occlusion of the latter.
- Obtain images (7 images in 14 sec) of the vertebrobasilar system using a rapid sequential angiography recording in a 5° left-anterior oblique position.
- Infiltrate the area around the right common femoral artery with local anesthetics (Lidocaine 1%, maximal 6 mg/kg). Make a small inguinal skin incision. Using the microscope for visualization, dissect the common femoral artery and secure with a proximal and distal ligature (4-0 polyfilament sutures) around the exposed end.
- After arteriotomy, cannulate the femoral artery with a 5-F sheath. Flush the side port of the sheath with saline.
- Advance a 5-F catheter into the brachiocephalic artery through the sheath under fluoroscopy. Create a road map, then advance a guide wire to the vertebrobasilar system. Inject a bolus of non-ionic Iopamidol (0.6 ml/kg, 5 ml/sec in 2 sec) for DSA as described in step 3.2.
4. Rotation to Prone Position
- Following baseline DSA, reposition the rabbit from supine to prone position. Be careful not to manipulate or relocate the position of the intra-arterial catheters.
- Position the head in a head-holder at a 30° angle, oriented head down.
5. Cisterna Magna Puncture
- Disinfect the skin over the head and neck with Povidone-iodine 3 times for 1 min each, and cover the surgical area with sterile sheets.
- Insert a 22 G x 40 mm pediatric spinal access needle transcutaneously into the cisterna magna without any prior skin incision or muscle displacement.
- Confirm that the animal is fully anesthetized by ensuring a lack of toe-pinch response before sliding the needle down along the bony external occipital protuberance until a gap is detected; do not push the needle any further.
- Confirm the correct positioning of the needle by observing spontaneous dripping of cerebrospinal fluid with the rabbit’s head tilted down at a 20° angle for a few min.
6. Installation of Intracranial Pressure and Cerebral Blood Flow Monitoring
- After midline skin and galea incision, insert a small surgical retractor.
- Make three round osteotomies (2 mm diameter) using a high-speed microdrill in the frontal part of the skull according to the outer skull landmarks (Figure 1)9, i.e., over the olfactory bulb and bilateral frontal for placement of a neuromonitoring device if necessary. Use a millimeter scale ruler to determine the coordinates for burr hole placement as follows: Intracranial pressure (ICP) monitoring in the midpupillary line, one to two mm from the midsagittal line; intraparenchymal laser-Doppler probes four to five mm anterior and lateral to the bregma (Figure 1).
- Visualize the dura, and perform meticulous hemostasis: use bone wax for bone hemostasis by virtue of its tamponade action and perform local hemostasis using bipolar coagulation of the dura.
- Place the intraparenchymal intracranial pressure (ICP) monitor tip into the right olfactory bulb to a depth of 2 mm and then calibrate.
- Place the two laser-Doppler flowmetry fine needle probes using an external clamp system and insert them into the corresponding burr holes in both the right and left frontal hemispheres lateral to the ventricular system, i.e., in the midline to avoid interference with cerebrospinal fluid. Place needle probes to a depth of 2.5 mm.
- After placement of the neuromonitoring probes, seal all burr holes with a thick plug of bone wax in order to keep the cranium fluid-tight.
- Measure baseline parameters of mean arterial blood pressure (MAP), ICP and cerebral blood flow (CBF)using a multiparameter monitor and four channel laser-Doppler tissue blood perfusion monitor.
7. Shunt Induction
- Connect the spinal access needle in the cisterna magna to the previously catheterized subclavian artery via blood-filled pressure monitoring tubing. Use the 3-way stopcock for blood pressure measurement and as blood sampling port.
NOTE: The severity of SAH depends on the amount of blood, and can be roughly estimated by the extensiveness of subarachnoid clots at the time of brain harvest5,11. - Continuously monitor MAP, HR, ECG, respiratory rate and end-tidal CO2 at a sampling rate of 1 Hz from 6 min before until at least 20 min after SAH.
- Confirm that the animal is fully anesthetized by ensuring a lack of toe pinch response before opening the shunt connection between the subclavian artery and the cisterna magna to induce SAH by the pressure gradient.
NOTE: A controlled SAH can be achieved by closing the shunt at any time point (e.g., at a desired level of ICP). - Record steady state values during a time period of approximately 15 min.
- After ICP reaches its peak, keep the spinal access needle in place until ICP returns to a steady state close to baseline values. If the ICP plateau is maintained for more than 10 sec or if ICP decreases spontaneously, close the shunt.
- Remove CBF fine-needle probes and ICP probe, plug burr holes with bone wax, remove all catheters (including subclavian catheter, since catheter manipulation with consecutive bleeding is associated with high morbidity and mortality, and increases infection rate), perform rigorous wound irrigation with Neomycin sulfate and suture the skin.
8. Postoperative Management
- The procedure lasts for about 2 hr. Due to the half-lives of ketamine and xylazine, the recovery time of the animal is quite short — approximately 1 hr. The animals are kept under a heating lamp during recovery. Additional fluids are not provided. During this initial postoperative recovery phase, apply buprenorphine 0.02 mg/kg s.c. every 8 hr for 24 hr.
- Apply transdermal fentanyl matrix patches releasing 12.5 μg/hr in the shaved neck region of the animals for effective analgesia over the next 72 hr.
- Do not leave an animal unattended until it has regained sufficient consciousness to maintain sternal recumbency.
- Do not return an animal that has undergone surgery in the company of other animals until fully recovered.
9. Follow up Digital Subtraction Angiography to Assess DCVS at Day 3
- Perform Steps 1.1 to 3.6 as described above.
- Euthanize the animals by intraarterial bolus injection of sodium thiopenthal (40 mg/kg) (Pentothal, Ospedalia AG, Hünenberg, Switzerland). In cases where histology and immunohistochemistry is needed, perform an intracardiac perfusion-fixation at RT at a perfusion pressure of 100 cm H2O.
Representative Results
The rabbit blood shunt model of SAH described in this report produces EBI in the hippocampus (Figure 2A, B), basal cortex (Figure 2A, B), and cerebral vasculature (Figure 2C) as early as 24 hr after injury and shows a characteristic blood distribution (Figure 2D)8. In addition, the model triggers moderate to severe degrees of DCVS on day three after SAH induction (Figure 3)10. The mortality rate is 20 – 30% due to respiratory arrest or severe bradycardia at the time of acute SAH. Almost all rabbits show progressive aggravation of neurological deficits from days 1 – 310. At that time point the animals are under complete anesthesia. From a technical point of view, the blood shunt model allows for both examiner-independent and controllable SAH induction (Figure 4).

Figure 1. Outer skull landmarks for CBF and ICP probes. (Reproduced with permission from the Journal of Neuroscience Methods 201:322 – 326) 9 Schematic drawing depicts the placement of intraparenchymal CBF and ICP probes in the frontal lobes and in the right olfactory bulb. CBF probes are placed 4 – 5 mm anterior and parasagittal to the bregma. ICP probes are placed in the midpupillary line in caudal–rostral direction at a distance of 1 – 2 mm from midsagittal line. Intraparenchymal CBF (Panels A and B) and ICP (Panels C and D) probes are shown in sagittal and coronal planes of T2-weighted MRIs. Note their relation to the ventricles. CBFl = left frontal burrhole for cerebral blood flow probes. CBFr = right frontal burr hole for cerebral blood flow probes. ICP = position of burr hole for intracranial pressure probes. *Bregma; **lambda. Please click here to view a larger version of this figure.
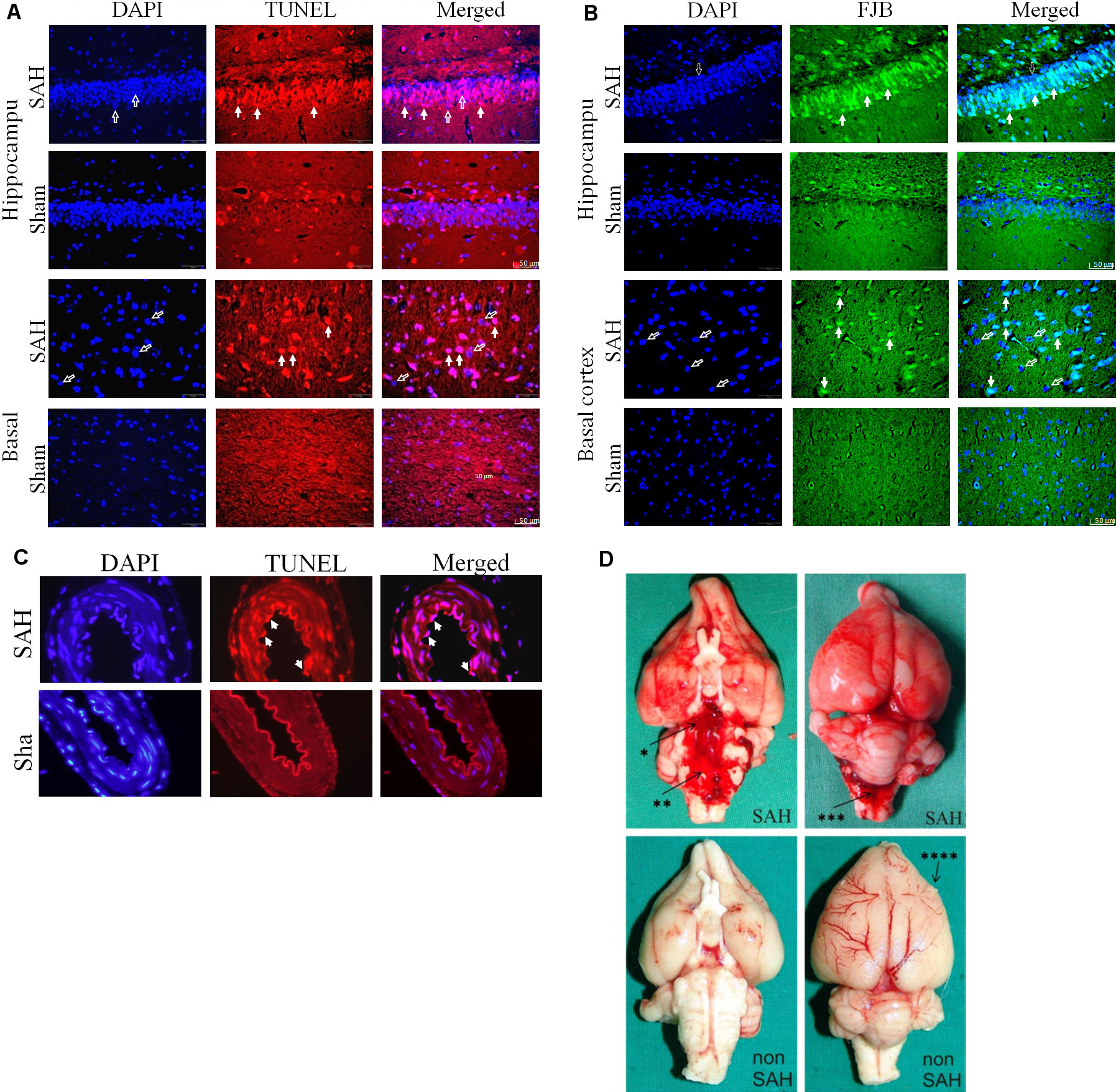
Figure 2. Early brain injury after subarachnoid hemorrhage. (Reproduced with permission from the Journal of Neuroscience Methods 208, 138 – 145 8 and the Journal of Neuroscience Methods 191:227 – 233) 10. TUNEL staining showed apoptosis in the hippocampus (A) and basal cortex (B) of SAH animals. Neurodegeneration was analyzed by FJB (Fluoro-jade stain) positive cells colocalized with DAPI. Colocalization with DAPI (left column) revealed that TUNEL positive staining (middle column) was localized in the nucleus (right column). Open arrows show DAPI positive nuclear staining. Solid arrows indicate TUNEL positive or FJB-positive cells. Scale bar = 50 μm. (C) Apoptosis and neurodegeneration are depicted 24 hr post SAH in basilar artery endothelial cells. Filled arrows indicate TUNEL- positive apoptotic, swollen, and detached endothelial cells. Scale bar = 50 μm. (D) Gross examination of the brain shows extended blood distribution on the ventral and dorsal surface of the brain and basal cisterns 24 hr after SAH induction as compared to sham-operated animals. * = basal cistern; ** = prepontine cistern; *** = cisterna magna; right frontal cortex lesion from the perioperative ICP monitor tip (**** = ICP probe lesion). Please click here to view a larger version of this figure.
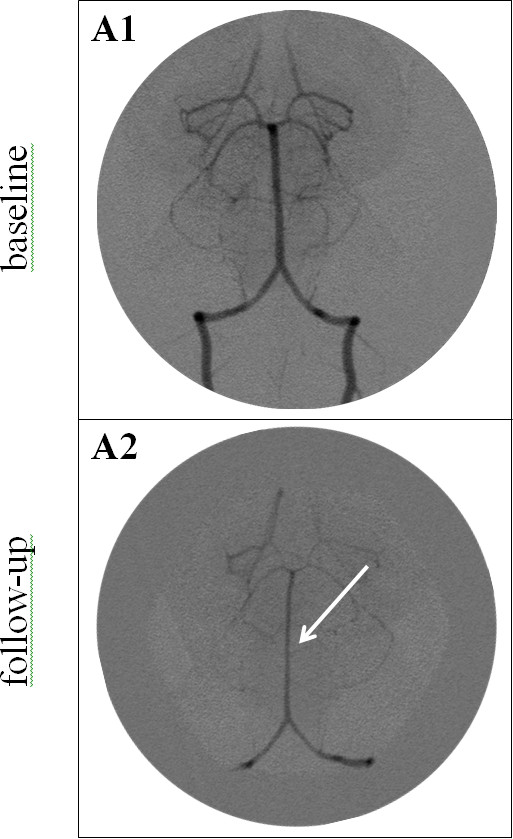
Figure 3. Vertebrobasilar angiograms before (A1) and after (A2) SAH induction. Baseline angiogram (A1) shows normal vessel diameter of the vertebral and basilar arteries. Three days after SAH induction (A2), angiogram shows a diffuse narrowing of the basilar (arrow) and vertebral arteries. Please click here to view a larger version of this figure.

Figure 4. ICP-controlled induction of various degrees of SAH. A key element of the rabbit blood shunt model is the ability to control for various degrees of SAH severity, including the amount of bleeding, an increase in intracranial pressure (ICP), or a reduction in cerebral perfusion pressure (CPP). Figure 4 illustrates the time course of the ICP following the induction of SAH (arrow = opening of the shunt). The curve progression depends on the physiology of the rabbit, primarily the pressure gradient between ICP and mean arterial blood pressure. If ICP reaches a value close to the diastolic blood pressure, the flow in the shunt stops. At that time point either ICP starts to decrease or the ICP value remains on a plateau. If the plateau stays longer than 10 sec the shunt is closed. Controlled SAH can be performed at any ICP level through closure of the shunt before spontaneous thrombosis (X = closure of the shunt). Please click here to view a larger version of this figure.
Discussion
The shunt model produces pathology similar to that observed in humans after acute SAH3,8,10. It has been suggested that EBI may exacerbate, maintain and even trigger DCVS12, and as such this model may aid in investigating both the early and late DCVS phases, including EBI and DCVS interactions following SAH. In particular, repeatable in vivo DCVS monitoring techniques including DSA13, computed tomography angiography14, and transcranial Doppler15 are more readibly applied in rabbits than in smaller laboratory animals. In addition to enabling examiner-independent spontaneous SAH, the model allows the user to control for different degrees of severity (amount of blood or ICP, and consequent changes in cerebral perfusion pressure).
Most importantly, this model results in a very consistent, reproducible, and pathophysiologically adapted course of events after acute SAH. Rebleeding is nonexistent and mortality is relatively low compared to other models of acute SAH16,17. Interposition of a flow probe in the shunt system further allows real-time assessment of SAH volume during bleeding10. However, the cost associated with large animal procedures is significantly greater than that of the more commonly used smaller laboratory animals, and genetic manipulation of rabbits is prohibitively challenging, thus limiting studies of particular genes on SAH outcomes18-20.
To ensure consistency and accuracy in the present model of SAH we recommend consideration of the following general criteria and critical surgical steps:
Perform experiments on three to four month old rabbits because i) the mortality rate after SAH induction is markedly reduced as compared to older rabbits, and ii) the vasospasm period is extended in older rabbits (20 to 40 months)21.
Calibration of the angiogram with an external sizing device (small sphere) allows assessment of vessel diameter in a blinded manner with low variability and accurate calibration at baseline and follow up angiograms. Measuring the vessel diameter three times (along a predefined length from the tip of the basilar artery) using automatic measurement analysis software to determine mean values (Step 3 in the protocol) is recommended.
Secure ligatures of the subclavian artery to prevent twisting or bending of the subclavian artery during repositioning from supine to prone position (Step 4 in the protocol).
Seal all burr holes with a thick plug of bone wax prior to SAH induction in order to keep the cranium fluid-tight and to main the steady state of ICP, CPP and MAP (Step 6 in the protocol).
Keep the spinal access needle in place until ICP returns to baseline values. Misplacement of the spinal access needle can result in significant morbidity (Step 7 in the protocol).
EBI and DCVS, both of which largely contribute to the unfavorable outcome and mortality after SAH, can be studied using the blood shunt model of SAH. Awareness of the individual technical details guarantees successful implementation of this model and allows for the evaluation of SAH sequelae and screening of potential treatment modalities.
Disclosures
The authors have nothing to disclose.
Acknowledgements
The authors thank Laurie von Melchner, Bern University Hospital, Department of Neurosurgery, Bern, Switzerland, for proofreading and editing the manuscript and Paskus Jeremiah, Boston Children’s Hospital, Boston, MA for proofreading the initial draft. We appreciate the skillful management of animal care, anesthesia, and operative assistance from Daniel Mettler, DVM, Max Müller, DVM, Daniel Zalokar, and Olgica Beslac, Experimental Surgical Institute, Department of Clinical Research, University of Bern, Bern, Switzerland. We thank Michael Lensch, Head Research Nurse, Department of Intensive Care Medicine, Bern University Hospital and University of Bern, Bern, Switzerland, for real-time data monitoring and post-processing of the physiological parameters. We thank Edin Nevzati, Carl Muroi, and Salome Erhardt, for their excellent laboratory technical and operative assistance.
This work was supported by the Department of Intensive Care Medicine, Bern University Hospital and University of Bern, Bern, Switzerland, the Department of Clinical Research, University of Bern, Bern, Switzerland, and the Research Fund from the Kantonsspital Aarau, Aarau, Switzerland. We thank Elsevier, for reprint permission for Figures 1 and 2.
Materials
| Name | Company | Catalog Number | Comments/Description |
| Equipment | |||
| operation microscope | Zeiss, Jena, Germany | Zeiss, OPMI-MD surgical microscope | |
| surgical equipment | B. Braun, Germany | forceps medical n°5, vessel sciccors 8cm, microclip 4mm | |
| respirator | Hugo Sachs | ||
| hair clipper | 3M Surgical Clipper | Starter Kit 9667A | |
| body warm plate | FHC | ||
| blood gas analyzer | Radiometer, Copenhagen, Denmark | ABL 725 | |
| cardiac monitoring | Camino Multi-Parameter Monitor, Integra, Plainsboro, NJ, US | AP-05 | |
| software analysis | BIOPAC Systems, Inc., Goleta, CA, USA | Biopac MP100 and acqKnowledge software,version 3.8.1 | |
| software analysis | ImagePro Discovery, MediaCybernetics, Silver Spring, MD, USA | image-Pro Plus version | |
| angiography apparatus | DFP 2000 A-Toshiba | MIIXR0001EAA | |
| ICP monitor | Camino Laboratories, San Diego, CA, USA | ICP monitor, Model 110-4B | |
| blood flow monitor | Oxford Optronix Ltd., Oxford, UK | CAL KIT microsphere solution | |
| laser-Doppler flowmetry fine needle probes | Oxford Optronix Ltd., Oxford, UK | MNP110XP, 0.48 mm diameter | |
| pressure tube | B. Braun, Germay | PE 1.0 mm × 2.0 mm | |
| anesthesia monitor | GE Medical Systems, Switzerland | Datex S5 Monitor | |
| Material | |||
| 20 G vascular catheter | Smiths Medical | Jelco i.v. catheter, REF 4057 | |
| 5.5F three-lumen central venous catheter | Connectors, Tagelswangen, Switzerland | silicone catheter STH-C040 | |
| 22Gx40mm needle | Emergo Group Inc., Netherlands | ||
| high-speed microdrill | Stryker, Solothurn, Switzerland | 5400-15 | |
| bone wax | Ethicon, Johnson & Johnson,NJ, USA | ETHW31G | |
| bipolar forceps | Aesculap, Inc., PA, US | US349SP | |
| Ketamin | Any generic product | ||
| Xylazine | Any generic product | ||
| Buprenorphine | Any generic product | ||
| Fentanyl | Any generic product | ||
| transdermal fentanyl matrix patches | Any generic product | ||
| Lidocaine 1% | Any generic product | ||
| 4% papaverin HCl | Any generic product | ||
| Neomycin sulfate | Research Organics Inc., OH, USA | Any generic product | |
| Povidone-iodine | Any generic product | ||
| 0.9% sodium chloride | Any generic product | ||
| Iopamidol | Abott Laboratories, IL, USA | Any generic product | |
| 3-0 resorbable suture | Ethicon Inc., USA | VCP824G | |
| 5-0 non absorbable suture | Ethicon Inc., USA | 8618G | |
| 4-0 polyfilament sutures | Ethicon Inc., USA | VCP284G | |
References
- Taylor, T. N., et al. Lifetime cost of stroke in the United States. Stroke; a journal of cerebral circulation. 27, 1459-1466 (1996).
- Kikkawa, Y., Kameda, K., Hirano, M., Sasaki, T., Hirano, K. Impaired feedback regulation of the receptor activity and the myofilament Ca2+ sensitivity contributes to increased vascular reactiveness after subarachnoid hemorrhage. Journal of cerebral blood flow and metabolism : official journal of the International Society of Cerebral Blood Flow and Metabolism. 30, 1637-1650 (2010).
- Marbacher, S., Fandino, J., Kitchen, N. D. Standard intracranial in vivo animal models of delayed cerebral vasospasm. British journal of neurosurgery. 24, 415-434 (2010).
- Marbacher, S., Neuschmelting, V., Graupner, T., Jakob, S. M., Fandino, J. Prevention of delayed cerebral vasospasm by continuous intrathecal infusion of glyceroltrinitrate and nimodipine in the rabbit model in vivo. Intensive care medicine. 34, 932-938 (2008).
- Zhou, M. L., et al. Comparison between one- and two-hemorrhage models of cerebral vasospasm in rabbits. Journal of neuroscience. 159, 318-324 (2007).
- Vatter, H., et al. Time course in the development of cerebral vasospasm after experimental subarachnoid hemorrhage: clinical and neuroradiological assessment of the rat double hemorrhage model. Neurosurgery. 58, 1190-1197 (2006).
- Lee, J. Y., Sagher, O., Keep, R., Hua, Y., Xi, G. Comparison of experimental rat models of early brain injury after subarachnoid hemorrhage. Neurosurgery. 65, 331-343 (2009).
- Marbacher, S., et al. A new rabbit model for the study of early brain injury after subarachnoid hemorrhage. Journal of neuroscience. 208, 138-145 (2012).
- Marbacher, S., et al. Outer skull landmark-based coordinates for measurement of cerebral blood flow and intracranial pressure in rabbits. Journal of neuroscience methods. 201, 322-326 (2011).
- Marbacher, S., et al. Extra-intracranial blood shunt mimicking aneurysm rupture: intracranial-pressure-controlled rabbit subarachnoid hemorrhage model. Journal of neuroscience. 191, 227-233 (2010).
- Sugawara, T., Ayer, R., Jadhav, V., Zhang, J. H. A new grading system evaluating bleeding scale in filament perforation subarachnoid hemorrhage rat model. J Neurosci Methods. 167, 327-334 (2008).
- Macdonald, R. L. Delayed neurological deterioration after subarachnoid haemorrhage. Nature reviews. Neurology. 10, 44-58 (2014).
- Zhang, Z. W., et al. Platelet-derived growth factor-induced severe and chronic vasoconstriction of cerebral arteries: proposed growth factor explanation of cerebral vasospasm. Neurosurgery. 66, 728-735 (2010).
- Laslo, A. M., Eastwood, J. D., Chen, F. X., Lee, T. Y. Dynamic CT perfusion imaging in subarachnoid hemorrhage-related vasospasm. AJNR. American journal of neuroradiology. 27, 624-631 (2006).
- Shao, Z., et al. Effects of tetramethylpyrazine on nitric oxide/cGMP signaling after cerebral vasospasm in rabbits. Brain research. 1361, 67-75 (2010).
- Bederson, J. B., Germano, I. M., Guarino, L. Cortical blood flow and cerebral perfusion pressure in a new noncraniotomy model of subarachnoid hemorrhage in the rat. Stroke; a journal of cerebral circulation. 26, 1086-1091 (1995).
- Veelken, J. A., Laing, R. J., Jakubowski, J. The Sheffield model of subarachnoid hemorrhage in rats. Stroke; a journal of cerebral circulation. 26, 1279-1283 (1995).
- Zakhartchenko, V., et al. Cell-mediated transgenesis in rabbits: chimeric and nuclear transfer animals. Biology of reproduction. 84, 229-237 (2011).
- Capecchi, M. R. Gene targeting in mice: functional analysis of the mammalian genome for the twenty-first century. Nature reviews. Genetics. 6, 507-512 (2005).
- Flisikowska, T., et al. Efficient immunoglobulin gene disruption and targeted replacement in rabbit using zinc finger nucleases. PloS one. 6, e21045 (2011).
- Nakajima, M., et al. Effects of aging on cerebral vasospasm after subarachnoid hemorrhage in rabbits. Stroke. 32, 620-628 (2001).

