Syntese av indoksyl-glykosider for deteksjon av glykosidase Aktiviteter
Summary
Indoxyl glycosides are well-established and widely used tools for enzyme screening and enzyme activity monitoring. Especially for glucose type structures previous syntheses proved to be challenging and low yielding. Our novel approach employs indoxylic acid esters as precious intermediates to yield a considerable number of indoxyl glycosides in good yields.
Abstract
Indoxyl glykosider viste seg å være verdifulle og allsidige verktøy for overvåking glykosidase aktiviteter. Indoxyls blir frigitt ved enzymatisk hydrolyse, og blir hurtig oksydert, for eksempel ved atmosfærisk oksygen, i Indigo typen fargestoffer. Denne reaksjonen muliggjør rask og enkel screening in vivo uten isolering eller rensing av enzymer, samt hurtigprøver på agarplater eller i løsning (for eksempel blå-hvit screening, mikro-brønner) og blir brukt i biokjemi, histokjemi, bakteriologi og molekylær biologi. Dessverre er syntesen av slike substrater viste seg å være vanskelig, på grunn av forskjellige bireaksjoner og den lave reaktiviteten av indoksyl hydroksylfunksjon. Spesielt for glukose typen strukturer lave utbytter ble observert. Vår ny tilnærming syssels indoxylic ester som viktige mellomprodukter. Indoxylic estere med varierte erstatningsmønstre var forberedt på skalerbare trasé. Faseoverførings glycosylations med disse acceptors og peracetylatedglycosylhalogenider kan utføres under vanlige betingelser i høye utbytter. Esterspalting og påfølgende mild sølv mediert glykosylering gir de peracetylated Indoxyl glykosider i høye utbytter. Endelig avbeskyttelse utføres ifølge Zemplén.
Introduction
For a long time the production of indigo was an economically very important process. Before large scale chemical syntheses gave cheap access to indigo, precursors were obtained from natural sources since pre-Christian times. The cultivation of indigo providing plants (natural indigo) in Europe became unrewarding in the 17th century, as the amount of indigo precursors of the Indian indigo plant (0.2-0.8 %) is about 30 times higher. At the end of the 19th century chemical synthesis of indigo suppressed the conventional cultivation1,2.
Indigo precursors occurring naturally in plants include Indican (1), Insatan A (2) and Isatan B (3) (Figure 1). All of them consist of an indoxyl motive linked to a glycosyl residue. Cleavage of the glycosidic linkage, for example by enzymatic hydrolysis, leads to release of indoxyl (4). Indoxyl itself is almost colorless, but can be rapidly oxidized to form an indigo dye (5). This sensitive reaction has been adapted in biochemistry, histochemistry, bacteriology and molecular biology for monitoring enzyme activities. Activity screening in vivo without isolation or purification of enzymes, as well as rapid tests on agar plates or in solution (e.g., blue-white screening, micro-wells) is possible. Depending of the residue (e.g., esters, glycosides, sulfates) linked to the indoxyl moiety, suitable substrates for different enzyme classes (e.g., esterases, glycosidases, sulfatases) have been developed3. In the following focus will be on formation and application of indoxyl glycosides.
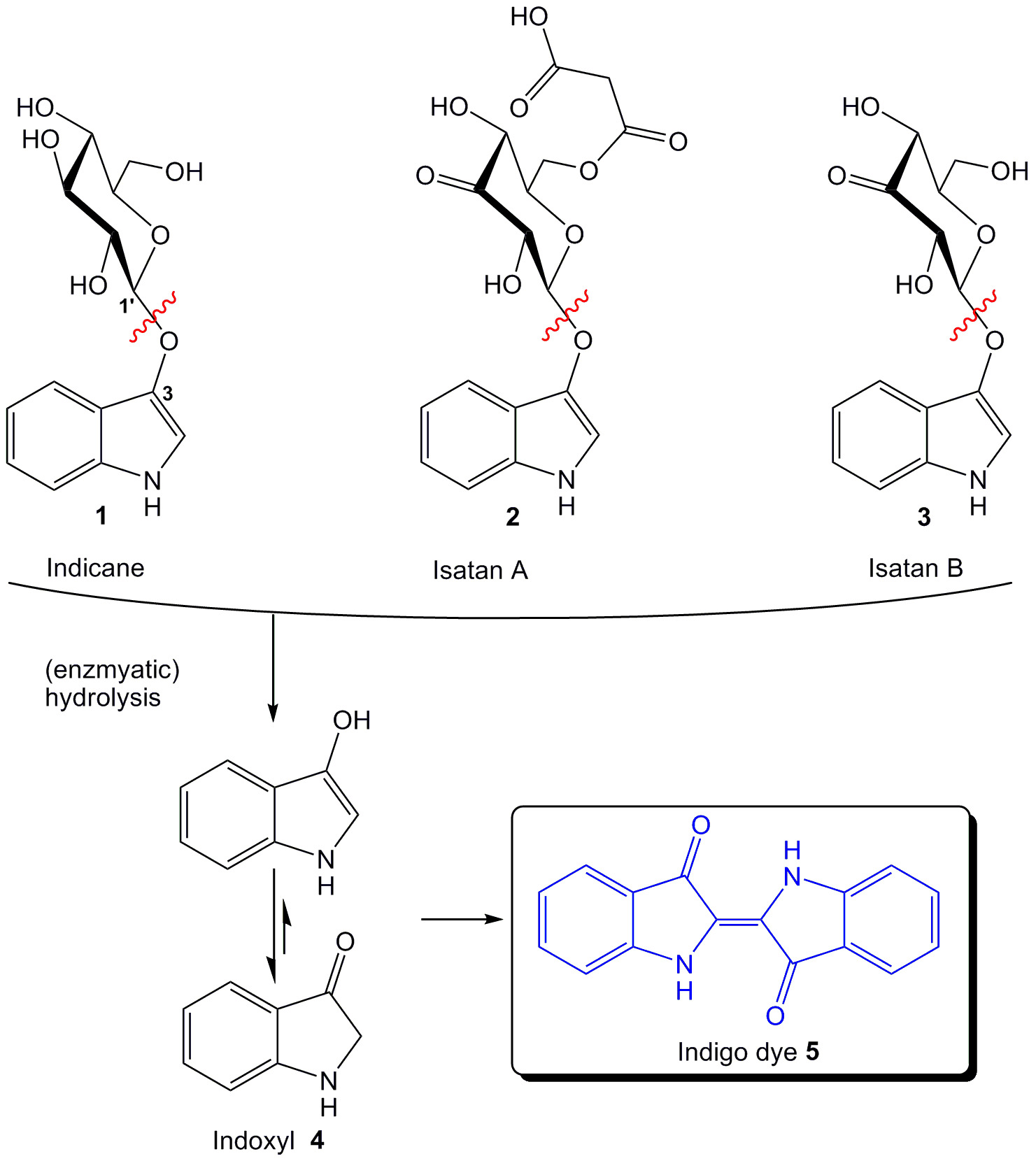
Figure 1: Natural indigo precursors and formation of indigo dye by hydrolysis. Please click here to view a larger version of this figure.
The substitution pattern of the indoxyl moiety determines the color and physical properties of the resulting indigo dye. The most common substitution patterns are 5-bromo-4-chloro (abbreviated by X; greenish-blue), 5-bromo (blue) and 5-bromo-6-chloro (magenta), since these form the smallest dye particles, do not form granules and have the least diffusion from sites of hydrolysis. The last property is especially important for in vivo experiments3.
The first report of an indigogenic method for detection of esterase activity was published in 1951 by Barrnett and Seligman, who employed indoxyl acetate and butyrate4. About one decade later the indigogenic principle was adapted for localization of mammalian glucosidase5. Up to now several indoxyl glycosides have been developed even though their synthesis proved to be difficult. Most syntheses are based on employing an N-acetylated indoxyl as acceptor and the respective glycosyl halide donor6-14. Glycosylation is performed in acetone with sodium hydroxide. Under these conditions a number of side reactions occur, decreasing the yield significantly. Especially for glucose type structures very low glycosylation yields were reported (e.g., 15% for (N-acetyl-5-bromo-4-chloro-indol-3-yl)-2,3,4,6-tetra-O-acetyl-β-ᴅ-glucopyranoside6 and 26% for (N-acetyl-5-bromo-4-chloro-indol-3-yl)-2,3,2',3',4'-penta-O-acetyl-β-ᴅ-xylobioside14 in a more recent example). Through a novel approach, employing indoxylic acid esters, a considerable number of indoxyl glycosides were prepared in good yields (e.g., (N-acetyl-5-bromo-4-chloro-indol-3-yl)-2,3,4,6-tetra-O-acetyl-β-ᴅ-glucopyranoside 57% yield).
The following protocol describes the straightforward synthesis of indoxylic acid allyl ester (5-bromo-4-chloro) and based thereon the synthesis of an indoxyl glycoside (X-Gal). A simple model experiment shows the enzyme reactivity of β-galactosidase employing X-Gal.
Protocol
Representative Results
Discussion
Owing to poor yields and limitations, especially for glucose type structures and more complex saccharides, a novel synthetic approach towards indoxyl glycosides was developed. Indoxylic acid esters proved to be precious key intermediates and were obtained in a modular, scalable pathway. All steps are high yielding and due to cheap starting materials and easy workup multi-gram syntheses are possible. The advantage of the allyl ester approach is the blocking of the reactive 2-position. Thus yield decreasing side reactions …
Disclosures
The authors have nothing to disclose.
Acknowledgements
Support of this work by Glycom A/S, Copenhagen, Denmark, is gratefully acknowledged.
Materials
| Name of Material/ Equipment | Company | Catalog Number | Comments/Description |
| Acetic anhydride | Grüssing | 10298 | Corrosive, flammable |
| Acetonitrile | Sigma-Aldrich | 608-001-00-3 | Harmful, flammable |
| Allyl alcohol | Aldrich | 453021 | Harmful, dangerous for the environment |
| Amberlite IR-120 H+ | Fluka | 06428 | Irritant |
| Bromoacetic acid | Merck | 802260 | Corrosive, toxic, dangerous for the environment |
| 4-Bromo-3-chloro-2-methylaniline | ABCR | AB 171687 | Irritant |
| Dichloromethane | ACROS | 326850010 | Harmful |
| Diethyl ether | Grüssing | 10274 | Harmful, extremly flammable |
| Dimethylformamide | ACROS | 348430010 | Harmful, flammable |
| Dimethylsulfoxide | Sigma-Aldrich | 41648 | |
| Ethyl acetate | Sigma-Aldrich | 607-022-00-5 | Irritant, flammable |
| Ethylenediaminetetraacetic acid | AppliChem | A1103.0500 | Irritant |
| ß1,3-Galactosidase, Recombinant, E. coli | Calbiochem | 345795 | |
| Hydrochloric acid | VWR | 20252.290 | Corrosive |
| Magnesium sulfate hydrate | Merck | 105885 | |
| Methanol | ACROS | 326950010 | Toxic, flammable |
| Morpholine | Janssen Chimica | 15.868.57 | Corrosive, flammable |
| Peroleum ether | Azelis | 111053 | Flammable, irritant, dangerous for the environment |
| Potassium carbonate | Grüssing | 12005 | Corrosive |
| Potassium permanganate | Grüssing | 12056 | Harmful, oxidising |
| Potassium tert-butoxide | Merck | 804918 | Corrosive, flammable |
| Pyridine | Sigma-Aldrich | 613-002-00-7 | Harmful, flammable |
| Silver acetate | Fluka | 85140 | Irritant, dangerous for the environment |
| Sodium bicarbonate | Grüssing | 12144 | Corrosive |
| Sodium hydride | Merck | 814552 | Corrosive, flammable |
| Sodium hydroxide | Riedel-de Häen | S181200 | Corrosive |
| Sodium methanolate | Merck | 806538 | Corrosive, flammable |
| Sodium sulfate | Grüssing | 12175 | |
| Tetrabutylammonium hydrogensulfate | Lancaster | 5438 | Harmful |
| Tetrahydrofurane | Sigma-Aldrich | 87371 | Harmful, flammable |
| Tetrakis(triphenylphosphine)palladium(0) | Sigma-Aldrich | 216666 | |
| Triphosgene | Fluka | 15217 | Toxic |
| Tris(hydroxymethyl)aminomethane hydrochloride | Sigma | T-3253 | Irritant |
References
- Clark, R. J. H., Cooksey, C. J., Daniels, M. A. M., Withnall, R. Indigo, Woad, and Tyrian Purple: Important Vat Dyes from Antique to the Present. Endeavour. 17, 191-199 (1993).
- Hunger, K. . Industrial Dyes: Chemistry, Properties, Applications. , (2003).
- Kiernan, J. A. Indigogenic Substrates for Detection and Localization of Enzymes. Biotechn. Histochem. 82, 73-103 (2007).
- Barnett, R. J., Seligman, A. M. Histochemical Demonstration of Esterases by Production of Indigo. Science. 114, 579-582 (1951).
- Pearson, B., Andrews, M., Grose, F. Histochemical Demonstration of Mammalian Glucosidase by Means of 3-(5-Bromoindolyl)-β-ᴅ-glucopyranoside. Exp. Biol. Med. 108, 619-623 (1961).
- Anderson, F. B., Leaback, D. H. Substrates for the Histochemical Localization of some Glycosidases. Tetrahedron. 12, 236-239 (1961).
- Van Dort, M. E., Lee, K. C., Hamilton, C. A., Rehemtulla, A., Ross, B. R. Radiosynthesis and Evaluation of 5-[125I]Iodoindolyl-3-yl-β-ᴅ-galactopyranoside ([125I]IBDG) as a β-Galactosidase Imaging Radioligand. Mol. Imaging. 7, 187-197 (2008).
- Yoshida, K., Iino, N., Koga, I. Syntheses of Halogen Substituted β-ᴅ-Glucuronides and Their Hydrolysis by Rabbit Liver β-Glucoronidase. Chem. Pharm. Bull. 32, 1759-1769 (1975).
- Horwitz, J. P., et al. Substrates for Cytochemical Demonstartion of Enzyme Activity I. Some Substituted 3-Indolyl-β-ᴅ-glycopyranosides. J. Med. Chem. 7, 574-575 (1964).
- Eschenfelder, V., Brossmer, R. 5-Bromo-indol-3-yl 5-Acetamido-3,5-dideoxy-α-ᴅ-glycero-ᴅ-galactononulopyranosidic Acid, a Novel Chromogenic Substrate for the Staining of Sialidase Activity. Glycoconjugate J. 4, 171-178 (1987).
- Fujii, I., Iwabuchi, Y., Teshima, T., Shiba, T., Kikuchi, M. X-Neu5Ac: A Novel Substrate for Chromogenic Assay of Neuraminidase Activity in Bacterial Expression Systems. Bioorg. Med. Chem. 1, 147-149 (1993).
- Berlin, W., Sauer, B. In situ Color Detection of α-ʟ-Arabinofuranisodase, a "No-Background" Reporter Gene, with 5-Bromo-3-indolyl-α-ʟ-arabinofuranoside. Anal. Biochem. 243, 171-175 (1996).
- Marmuse, L., et al. New Chromogenic Substrates for Feruloyl Esterases. Org. Biomol. Chem. 6, 1208-1214 (2008).
- Kaneko, S., Kiaoka, M., Kuno, A., Hayashi, K. Syntheses of 4-Methylumbelliferyl-β-ᴅ-Xylobioside and 5-Bromo-3-Indolyl-β-ᴅ-Xylobioside for Sensitive Detection of Xylanase Activity on Agar Plates. Biosci. Biotechnol. Biochem. 64, 741-745 (2000).
- Robertson, A. J. Syntheses of Glucosides. Part I. The Synthesis of Indican. Chem. Soc. , 1937-1943 (1927).
- Robertson, A., Waters, R. B. J. Synthese of Glucosides. Part VII. The Synthesis of 6-Bromoindican. Chem. Soc. , 2239-2243 (1929).
- Böttcher, S., Thiem, J. Indoxylic Acid Esters as Convenient Intermediates Towards Indoxyl Glycosides. Eur. J. Org. Chem. , 564-574 (2014).
- Böttcher, S., Hederos, M., Champion, E., Dékány, G., Thiem, J. Novel Efficient Routes to Indoxyl Glycosides for Monitoring Glycosidase Activities. Org. Lett. 15, 3766-3769 (2013).
- Böttcher, S., Thiem, J. Facile Preparation of Indoxyl- and Nitrophenyl Glycosides of Lactosamine and Isolactosamine. RSC Adv. 4, 10856-10861 (2014).

