Chapter 12
Solutions and Colloids

There is no one solvent that can dissolve every type of solute. Some substances that readily dissolve in a certain solvent might be insoluble in a…

The formation of a solution is an example of a spontaneous process, a process that occurs under specified conditions without energy from some…

Water and other polar molecules are attracted to ions. The electrostatic attraction between an ion and a molecule with a dipole is called an…

Imagine adding a small amount of sugar to a glass of water, stirring until all the sugar has dissolved, and then adding a bit more. You can repeat…

A solute is a component of a solution that is typically present at a much lower concentration than the solvent. Solute concentrations are often…

The equilibrium vapor pressure of a liquid is the pressure exerted by its gaseous phase when vaporization and condensation are occurring at equal…
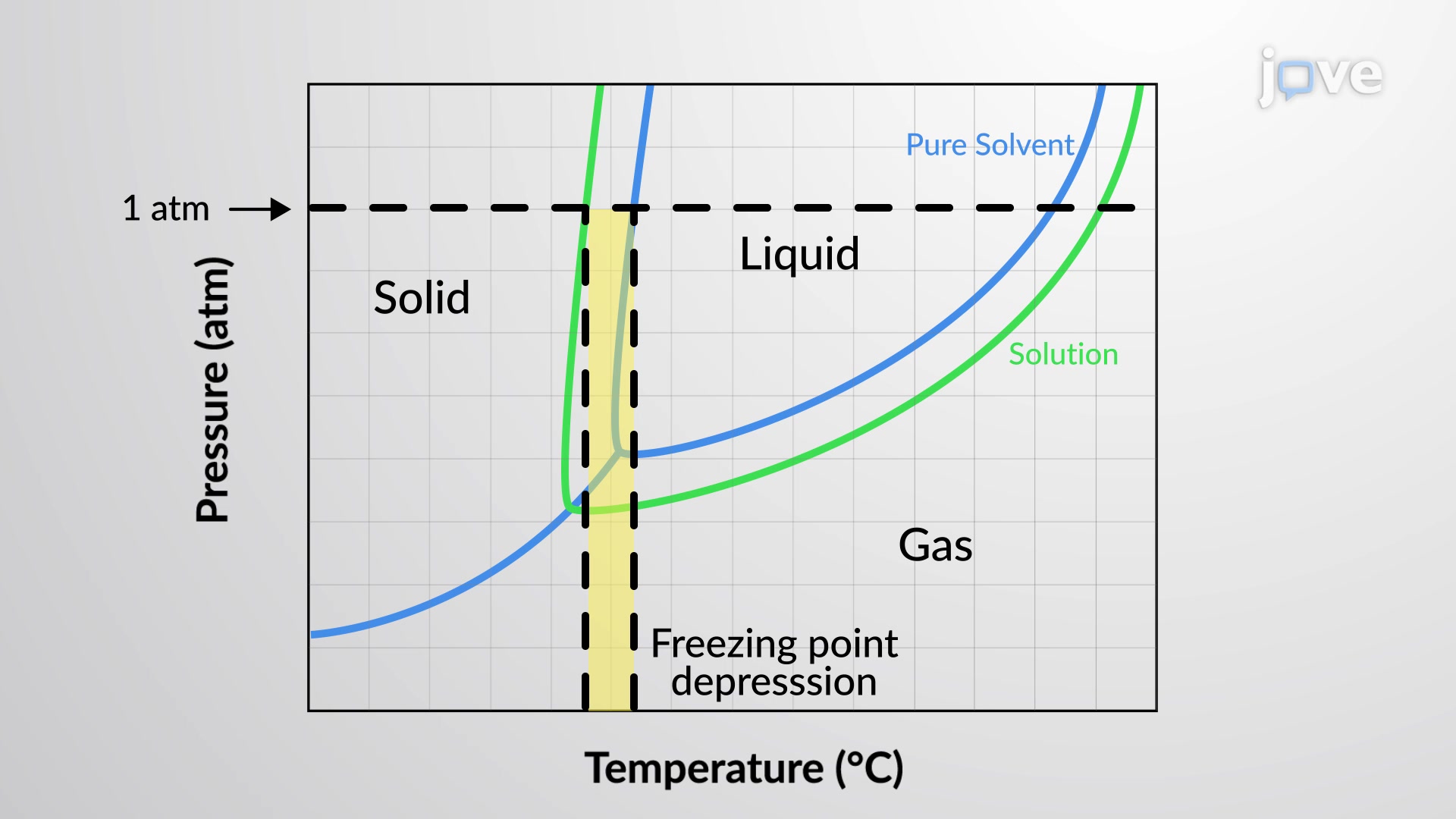
Boiling Point Elevation
The boiling point of a liquid is the temperature at which its vapor pressure is equal to ambient atmospheric pressure. Since…

A number of natural and synthetic materials exhibit selective permeation, meaning that only molecules or ions of a certain size, shape, polarity,…

Fundamentals of inorganic-organic interactions are critically important in the discovery and development of novel biointerfaces amenable for…

Control over colloidal assembly is of utmost importance for the development of functional colloidal materials with tailored structural and mechanical…








