컬럼 크로마토그래피를 이용한 총 지질 추출물의 정제
English
Share
Overview
출처: 제프 살라컵 연구소 – 매사추세츠 대학교 애머스트
유기 용매 추출의 제품, 총 지질 추출물 (TLE), 종종 수백의 복잡 한 혼합물, 하지 않을 경우 수천, 다른 화합물의. 연구원은 종종 화합물의 소수에 만 관심이. 관심 있는 화합물은 알칸, 케톤, 알코올 또는산(도 1)과같은 여러 화합물 클래스 중 하나에 속할 수 있으며, 관심 있는 화합물을 보다 명확하게 보기 위해 속하지 않는 화합물 클래스를 제거하는 것이 유용할 수 있다. 예를 들어, TLE는 1,000개의 화합물을 함유할 수 있지만, Uk’37 해표면 온도 프록시는 2개의 화합물(alkenones)과 TEX86 해표면 온도 프록시만을 기반으로 하며 4개(글리세롤 글리세롤 테트라에테르)에 만 기초한다. 그것은 그들이 에 관심이없는 화합물의 많은 제거하기 위해 연구원을 behoove 것. 이 관심의 화합물의 기악 분석 (알케네네스 또는 GDGTs) 덜 다른 외부 화합물에 의해 복잡 하 게 될 가능성이.
다른 경우에, 업스트림 정제 기술은 우리의 이전 비디오에서 처포온화 도중 카복실산의 생산과 같은 견본에서 지금 제거하려는 화합물을 생성할 수 있습니다. 위의 두 경우 모두 컬럼 크로마토그래피라고 하는 정화 기술이 매우 유용합니다.
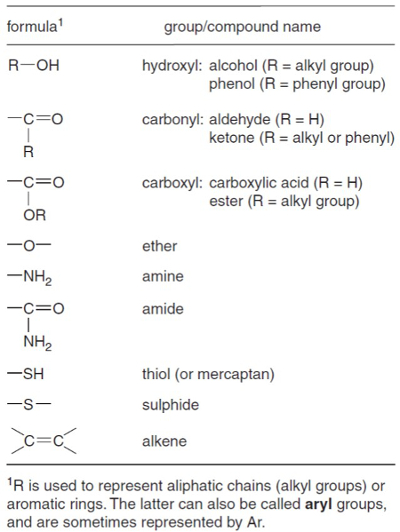
그림 1. 지구화학적으로 중요한 기능 성 그룹. 킬롭스와 킬롭 스1에서.
Principles
Procedure
Results
This purification technique produces three different vials, each containing a different compound class or group of compound classes. The complexity of any sample to be analyzed on an instrument has been vastly decreased. This process also removes compounds, such as acids produced during a saponification, that can actually stick to parts of the instruments, because of their low volatility, which would decrease their accuracy, precision, and lifetime.
Applications and Summary
Alkenones and isoprenoidal GDGTs are both very common constituents of marine sediments and can be found across the world's oceans. Alkenones are being increasingly detected in lake sediments, although the organisms responsible for their production are different than in the ocean, and thus the relationship between the Uk'37 ratio and water temperature (calibration) is different from the ocean and even between separate lakes. Isoprenoidal GDGTs are found in some large lakes and just like alkenones, often need a local calibration.
The alkenones and GDGTs we are interested in come out in the ketone and polar fractions, respectively. In marine sediments we often analyze both sea surface temperature (SST) proxies from one sample. This allows the construction of two independent SST records, which show the evolution of water temperature at the core site through time. This comparison, called a multi-proxy approach, often highlights times when the two proxies agree and times when they don't. This agreement or discrepancy itself contains information. If the two proxies agree, maybe the producing organisms occupied the same depth habitat, or maybe they lived at separate depths but a well-mixed water column led to the vertical homogenization of temperature (water usually cools with depth). If the two proxies disagree, it could be that the two populations lived at separate depths; one living in warm, shallow waters and one in cooler, deeper water. Or it could be that the compounds were produced during different times of the year and so reflect the temperatures of different seasons. These questions are created by the analysis of two different SST proxies at the same site and they highlight the care organic geochemists and paleo-climatologists need to take when interpreting their data.
Because of the high relative stability of apolar hydrocarbons, the apolar fraction contains many interesting organic compounds. Alkanes are important constituents of a leaf's outer waxy layer and they are used in sediment records for many reasons. Their average chain length (number of carbon atoms) contains information on the dominance of aquatic vs. terrestrial plants, temperature, and precipitation. The isotopic ratio of carbon in alkanes is related to the C3 vs. C4 plant-type of the plant that produced it and the hydrogen isotopic ratio is related to local to global temperature and precipitation. Steranes and hopanes are also found in the apolar fraction. These biomarkers are the geostable versions of bioactive compounds like hopanoids and steroids, which serve important biochemical roles in prokaryotes and eukaryotes, respectively.
The mid-polarity fraction contains our alkenones. Alkenones are ketones, which are important recorders of ancient surface temperatures via the Uk'37 sea surface temperature proxy. Some ketones also come from the same leaf waxes the alkanes do, although there are generally far less.
The polar fraction contains carboxylic acids, another important constituent in leaf wax, that is slightly less specific and harder to work with than alkanes (low volatility) but can nonetheless relate some of the same information. Glycerol dialkyl glycerol tetraethers (GDGTs) are in the polar fraction and are another important recorder of ancient water and air temperatures.
References
Killops, S. and Killops, V. Introduction to Organic Geochemistry. Blackwell Publishing, Malden, MA (2005).
Transcript
Column chromatography is a flexible technique for purifying the complex mixture of compounds found in sediment. Mixtures separate as they move through the column and are collected in fractions, each containing a different chemical class of compounds. Therefore, column chromatography is often used as an additional purification step after initial isolation of the desired compound. Organic extracts such as total lipid extracts may be complex mixtures of many compounds. Some purification techniques, such as saponification, introduce compounds that can damage analytical instruments and so must be removed before analysis. This video is part of a series on lipid extraction, purification, and analysis from sediments. Once a total lipid extract is collected from a sedimentary sample, column chromatography is used to purify both alkenones and GDGTs, depending on the desired analysis.
In column chromatography, a mixture of chemical compounds is loaded onto a solid stationary phase such as silica gel. A mobile phase such as an organic solvent is then used to elute, or remove, the compounds from the column. The order in which the compounds are eluted depends on the strength of the interactions of the compounds with the silica gel and with the eluent.
The eluate is collected in fractions, each containing different compounds from the mixture. Depending on the properties of the compounds, a single solvent may provide sufficient separation and elute all of the compounds of interest. Otherwise, multiple solvents are used to elute each compound of interest in turn.
Polar compounds, which have an uneven distribution of charge, adsorb strongly to the polar silica gel, whereas apolar compounds adsorb weakly. Polar solvents have greater affinity for silica gel and therefore are more powerful eluents than apolar solvents. Thus, apolar solvents elute only apolar compounds, whereas polar solvents elute both apolar and polar compounds.
When the desired compounds are moderately polar, apolar compounds should be washed off the column with an apolar solvent before a polar solvent is used. To avoid eluting unwanted highly polar compounds such as acids, a polar eluent should not have more eluting power than needed for the most polar desired compound.
Now that you understand the principles of column chromatography, let’s go through a procedure for purification of lipid biomarkers from a total lipid extract by silica gel column chromatography.
To remove organic contaminants, combust borosilicate glass pipettes, borosilicate glass vials, and glass wool for 6 h at 550 °C. Once the glassware is ready for use, set up a rack to hold pipettes and vials. Obtain pipette bulbs, clean tweezers, a laboratory spatula, silica gel, hexane, dichloromethane, and methanol. With clean tweezers, place a small tuft of glass wool into the mouth of a pipette. Gently push the glass wool to the bottom of the pipette with the stem of another pipette to form a loose plug. Carefully load silica gel into the pipette until half full. Secure the pipette upright in the rack. Secure a 4-mL borosilicate glass vial below the tip of the pipette for waste collection. In another borosilicate glass vial, suspend up to 10 mg of dry sample from the saponification process in hexane. If the sample sticks to the walls of the vial, sonicate the vial for 5 min. The chromatography procedure can now begin.
To begin the chromatography, wash the silica gel column with 3 volumes of hexane to remove air bubbles and impurities. Then, replace the waste vial with an empty vial for the apolar fraction. With a glass pipette, load the sample onto the column and allow the suspension to soak into the silica gel. Work quickly so the column does not dry out during the procedure. Rinse the sample vial twice with small portions of hexane and transfer each rinse to the column. Continue adding hexane to the column until the collection vial is nearly full. Allow all of the hexane to finish entering the silica gel. Then, exchange the filled vial with an empty vial for the mid-polar fraction. Next, rinse the sample vial with DCM and add it to the column 3 times. Continue adding DCM to the column until the collection vial is nearly full. Allow the DCM to finish soaking into the silica gel and then exchange the filled vial with an empty vial for the polar fraction. Repeat this process with methanol.
Once the vial is nearly full, allow the methanol to finish dripping into the vial and then cap all of the vials. The mid-polar fraction contains the desired alkenones, while the GDGTs are in the polar fraction. For particularly dirty or complex alkenone samples, the mid-polar fraction must be further purified with urea adduction, before analysis.
Column chromatography is widely used in chemistry as an analytical and purification technique.
Carbon nanotubes, or CNTs, are increasingly used in many industries, but there is growing concern of their effects on human health. Varying the properties of CNTs changes how they behave in water and soil. To investigate how well porous media like sand and dirt retain CNTs, a column was prepared with porous soil as the stationary phase. First, fractions were collected during loading of the CNT solution onto the column to analyze the transport of CNTs through soil. Then, the CNTs still adsorbed to the soil were eluted and the fractions analyzed for the amount of CNTs that had remained in soil. The results lend insight into the relationship between CNT surface functionalization and their transport mechanisms in the environment.
Column chromatography can be operated on both large and small scales, and is therefore used when designing syntheses for industrial applications. Spider silks have excellent tensile strength and ductility, but cannot be harvested on an industrial scale. Following silk protein synthesis, the recombinant silk proteins are purified by affinity chromatography, in which the stationary phase is designed to trap only the desired molecule. Thorough washing and elution grants the pure protein fractions needed to spin spider silk in large scales.
Many stationary phases are available for column chromatography. One stationary phase may not be suitable for all potential products of a synthesis with a broad substituent scope, such as this iodoaziridine synthesis. Crude product is mixed with various stationary phases and decomposition assessed by proton NMR. As proton NMR is highly sensitive, many stationary phases can be screened for product decomposition using a small amount of crude product. Column chromatography is then performed with the optimal stationary phase, allowing purification of novel and highly reactive compounds.
You’ve just watched JoVE’s introduction to column chromatography for the purification of a total lipid extract. The following video will demonstrate how to further purify complex mixtures containing alkenones.
Thanks for watching!
