Non-fluoroscopic Catheter Tracking for Fluoroscopy Reduction in Interventional Electrophysiology
Summary
Radiation exposure is an underestimated risk in complex ablation procedures. Here, we describe a protocol to significantly decrease fluoroscopy time and dosage for both the patient and the lab staff by using a novel non-fluoroscopic catheter visualization system.
Abstract
A technological platform (MediGuide) has been recently introduced for non-fluoroscopic catheter tracking. In several studies, we have demonstrated that the application of this non-fluoroscopic catheter visualization system (NFCV) reduces fluoroscopy time and dose by 90-95% in a variety of electrophysiology (EP) procedures. This can be of relevance not only to the patients, but also to the nurses and physicians working in the EP lab. Furthermore, in a subset of indications such as supraventricular tachycardias, NFCV enables a fully non-fluoroscopic procedure and allows the lab staff to work without wearing lead aprons. With this protocol, we demonstrate that even complex procedures such as ablations of atrial fibrillation, that are typically associated with fluoroscopy times of >30 min in conventional settings, can safely be performed with a reduction of >90% in fluoroscopy exposure by the additional use of NFCV.
Introduction
Catheter ablation has become a standard therapy in the treatment of many arrhythmias. While different ablation strategies have been proposed and are currently applied, all ablation procedures share one commonality in their necessity for the use of fluoroscopy to visualize catheters. Heavy reliance on the use of live x-ray for ablation procedures was alleviated in the 1990s with the advent of 3D electroanatomical mapping systems (EAMS) that helped to significantly reduce radiation time and dosage. Integration of cardiac imaging using magnetic resonance imaging (MRI) and computed tomography (CT) was shown to even further reduce fluoroscopy exposure during ablation procedures1. More recently, a new technology for catheter visualization, called the MediGuide-(MG) technology, has been introduced that can further facilitate reduction in radiation exposure2,3. Details have been previously described4,5. Briefly, single-coil sensors embedded in the catheter tip can be accurately localized by an electromagnetic field. Information about the 3D position and orientation of the tools is then transferred to the fluoroscopy system and is used to visualize the catheter tip in a virtual bi-planar view projected on 2 pre-recorded cine loops. It has been previously shown that the application of the MG technology can lead to a significant reduction in fluoroscopy burden by using diagnostic catheters in atrial flutter4 and by using both diagnostic and ablation catheters in several supraventricular tachycardias (SVT)6 and atrial fibrillation (AF)7 cases. There may be concerns that the application of the non-fluoroscopic catheter visualization (NFCV) technology may increase procedural risks in the absence of the catheter shaft visualization and catheter localization that is solely based on the location of the catheter tip. It was demonstrated that the complication rate is equal or even lower to procedures performed with conventional tools14. This could be explained by a limitation of conventional procedures: only in a certain percentage of the procedure catheters will be “visible”. This changed by application of NFCV technology since catheters will be visible during the entire procedure on this virtual biplanar view.
In this protocol, we perform an ablation of atrial fibrillation in a patient with paroxysmal, drug-refractory and highly symptomatic atrial fibrillation. The goal of this protocol is to achieve the same endpoints as in a conventional procedure, i.e., isolation of all pulmonary veins with proven bi-directional block, and to reduce fluoroscopy exposure for the patient by >90% as compared to conventional settings via the additional use of the NFCV technology.
Protocol
All patients signed an informed consent form after all typical complications of an ablation procedure such as pericardial effusion, vascular complications at the access site, stroke/TIA, and esophago-atrial fistula, were explained. This fulfilled the requirements of the local ethics committee. No patient subgroup had to be excluded (e.g., patients with pacemakers or ICD); only general contraindications for AF ablation procedures (e.g., contraindication for anticoagulation, hyperthyreosis, valvular AF, etc.) had to be addressed.
1. Patient Setup
- On the day of hospital admission, perform a routine physical exam including resting ECG, blood analysis, transesophageal echo and a contrast-enhanced CT scan of the heart on the patient. International normalized ratio (INR) should be between 2 and 3.
- If novel anticoagulants are used, skip either 1 dose (rivaroxaban) or 2 doses (dabigatran and apixaban) before the procedure.
2. Ablation Procedure
- Place the 3D EAMS patches on the patient per manufacturer’s instructions on the thorax (front and back; left and right), neck, and belly. Monitor oxygen saturation using a finger clip, as well as the non-invasive blood pressure. Perform disinfection of the groin region.
- Administer midazolam (2-3 mg, i.v.) and fentanyl (0.025 mg, i.v.) as premedication to slightly sedate the patient and to provide some analgesics during the puncture of the femoral vessels.
- Start the procedure with the injection of 40 ml of 1% Mepivacain to the left and right groin areas.
- Perform punctures of the femoral vessels. Start the puncture for venous access 1 cm medial to the femoral artery, 1 cm below the connection between symphysis and crista iliaca anterior superior. Perform the arterial puncture 1 cm superior to the venous access sites.
- Perform two 7F punctures in the left femoral vein for placing two diagnostic catheters: 1 steerable decapolar catheter for the coronary sinus (CS) and 1 steerable decapolar catheter for the right ventricular apex. After a successful puncture of the vessels, advance a guidewire, remove the puncture needle and place the sheath over the wire per the Seldinger technique.
- Next, perform 2 punctures on the right side: a 4F puncture in the right femoral artery for invasive blood pressure measurements, and an 11F one in the right femoral vein for the transseptal sheath. Before inserting the 11F sheath, control the intravasal position of the wire using fluoroscopy and then place the sheath.
- Administer heparin (100 IU/kg, i.v.) for anticoagulation.
- Check the activated clotting time (ACT) every 20 min; the target for the anticoagulation is an ACT between 250 and 350 sec. If necessary, administer boluses of heparin according to ACT measurements.
- During the procedure, maintain the patient in a deep analgosedation using midazolam (2-5 mg), fentanyl (0.05-0.1 mg) and propofol (bolus 0.5 mg/kg and constant basal rate of 0.5 mg/kg/h).
- Acquire 2 live fluoroscopy or cine loops using the X-Ray fluoroscopy system in a right anterior oblique projection (RAO 15°) and a left anterior oblique projection (LAO 50°), each approximately 3 sec long (Figure 3 and Video 1)7,14,15.
NOTE: The NFCV projects the catheter tips on these pre-recorded cine loops allowing a non-fluoroscopic placement of the diagnostic catheters. - Place one of the diagnostic catheters in the CS by first advancing the catheter tip to the superior vena cava (SVC), then pulling it back slowly and deflecting it to bring it close to the bundle of His.
- Deflect the catheter to its maximum allowable curve and perform a clockwise rotation to bring the tip to the CS ostium. Advance the catheter as deep as possible in the CS to achieve a stable position. Then, place a landmark on the catheter tip using the NFCV system to mark the location.
- Use the other diagnostic catheter to place landmarks for superior vena cava (SVC), inferior vena cava (IVC) and fossa ovalis (see Figure 1).
- Perform a trans-atrial septal puncture using a long steerable sheath8.
- Insert a long guidewire to the SVC and verify the position with fluoroscopy. Advance the steerable sheath over the wire to the junction between the SVC and the right atrium (RA). Insert a long needle into the dilator, pull back the sheath until it “jumps” into the fossa ovalis.
- Perform the puncture by advancing the needle and injecting contrast dye (15 ml of Ultravist 300) to verify the correct position of the sheath in the left atrium (LA).
- Once the needle tip is in the LA, advance the dilator to LA, disconnect it from the sheath and advance the sheath over the dilator into the LA. Deflect the sheath and slowly remove the needle and dilator from the sheath.
- Aspire 10 ml of blood from the sheath and carefully flush the sheath with heparinized saline. Flush the sheath with heparinized saline constantly at a flow rate of 2 ml/hr.
NOTE: The diagnostic catheter is used for electroanatomical reconstruction of the LA and pulmonary venous anatomy. Coronary sinus catheter remains in place and serves as the reference catheter for the 3D mapping system.
- Insert the long sheath in the superior pulmonary veins and perform 2 new fluoroscopy or cine loops in RAO 15° (right PV) and LAO 50° (left PV) during injection of 15 ml of contrast dye (Ultravist 300)7,14,15.
- Perform fusion of the electroanatomical map with 3D reconstructed CT anatomy. Map anatomical landmarks in the LA carefully and use for this co-registration process.
- For example, use the junction of the left inferior pulmonary vein (LIPV) to the LA body. Take at least 10 – 15 points for the fusion process and then double-check and optimize the co-registration process with the roving catheter. Upon completion, the segmented CT model is positioned at an anatomically correct position in 3D space.
- Place a temperature probe with 3 thermocouples trans-orally to measure the intra-luminal intra-esophageal temperature at the level of the LA.
- Insert the ablation catheter via the transseptal sheath.
- Perform ablation around the ipsilateral pulmonary veins by using power settings of 35 W (anterior) and 25 W (posterior) at an irrigation rate of 17 ml/min.
- If the intra-luminal temperature exceeds 39 °C, immediately stop the ablation and adjust the power settings, lowering the minimum power to be 20 W. In case of temperature rise despite reduction of energy settings, consider modifying the lesion setting to go more antral if it is too close to the esophagus.
- If the temperature still exceeds 41 °C, perform an esophagoscopy the day after the ablation to exclude mucosal thermal damage which could develop to an esophago-atrial fistula10.
- Check the completeness of the pulmonary vein isolation using a decapolar circular catheter by pacing maneuvers at maximum power (usually 10 mA/msec) from all bipoles of the spiral catheter. Make sure that the stimulus does not capture the LA by checking signals on the CS catheter. Verify that there is no LA capture in any of the pulmonary veins.
- If necessary, detect and close “gaps” in the lesion set: move the ablation catheter around the circumferential lesions and stimulate with maximum output from the tip of the ablation catheter. If the atrium is captured, start ablation until the local capture disappears16. Use this “pace-and-ablate”-technique around all pulmonary veins.
- Once the isolation line around the pulmonary veins is completed, perform a voltage map of the left atrium to determine a healthy atrium (shown in purple) or a fibrotic atrium (shown in blue, yellow, and grey) [see Figure 1 and 2]17. Use the cut-off values 0.5 mV for normal tissue and 0.2 mV for scar tissue17.
- Use the ablation catheter or the diagnostic catheter and start in the pulmonary vein antrum. Make sure that there is sufficient contact with the catheter tip and register the local signal amplitude to the 3D mapping system. For a normal sized LA, take points covering the entire LA body and PV antrum (100-150 points).
- Perform a test for inducibility with 20 sec burst-pacing from the coronary sinus with cycle lengths of 300 msec, 250 msec and 200 msec or to atrial refractory time. If a stable atrial tachycardia or atrial flutter is induced, map and ablate accordingly.
- If atrial fibrillation is induced, perform an electrical cardioversion (with 200 J biphasic shock) and terminate the procedure.
- Remove the transseptal sheath and catheters.
- Antagonize heparin by injecting protaminsulfat (10,000 IU, i.v.) and remove the sheaths from the groin.
3. Post-procedural Management
- Perform 10 min of manual femoral compression. After removal of the sheaths manually, compress the puncture sites on both sides. Check if there is still active bleeding after 5 min. If not, continue compression for at least an additional 5 min and place the pressure bandage for 6 hr.
- Subject the patient to 6 hr of bed rest before the patient is remobilized.
- After removal of the pressure bandage and checking the femoral vessels clinically (palpation and auscultation), deliver the next dose of anticoagulant drugs (warfarin or novel anticoagulants).
Representative Results
This procedure typically lasts 2-2.5 hr. The patients are under deep analog-sedation, meaning that they are sleeping, receiving analgesics but breathe spontaneously. If all endpoints including bi-directional block in all pulmonary veins, healthy left atrial tissue, and non-inducibility of atrial fibrillation or atrial flutter are achieved, patients have about a 75% probability of freedom from atrial fibrillation recurrence after 12 months. If the left atrium has fibrotic tissue with low voltage areas (see Figure 2), the chances of permanent freedom from arrhythmias decrease compared to patients with healthy left atrial tissue (see Figure 3). Typically, patients can be discharged 24 hr after the procedure. In the first 4-6 weeks after the ablation procedure, short episodes of atrial arrhythmias can occur and are frequent. After 6 weeks, the likely outcomes of the ablation procedure are evident. In most cases, all medical anti-arrhythmic treatments are discontinued on the day of the ablation procedure. Oral anticoagulation is mandatory and needs to be continued after the ablation procedure irrespective of the individual’s stroke risk for at least 3 months.

Figure 1: Ablation of atrial fibrillation using NFCV technology. Left and middle: catheter visualization using the NFCV technology: ablation catheter (red tip) in the left superior pulmonary vein (LSPV, blue marker). Right: the same setting displayed in the 3D mapping system. Ablation catheter (green halo) placed in the left superior pulmonary vein close to the ridge to the left atrial appendage. Esophageal temperature probe posterior to the left atrium (green catheter). Please click here to view a larger version of this figure.

Figure 2: Voltage map of a “diseased” left atrium. 3D reconstructed CT with low-voltage areas at the posterior wall of the left atrium and in the mitral isthmus region indicating areas of previous ablation. Please click here to view a larger version of this figure.

Figure 3: Voltage map of a “healthy” left atrium. 3D reconstructed CT model of a left atrium. A color-coded voltage map is shown with purple for healthy tissue (electrograms >0.5 mV) and grey for scar tissue (electrograms <0.2 mV). Electrogram amplitudes >0.2 mV and <0.5 mV are displayed in yellow, red, and blue. Please click here to view a larger version of this figure.
Video 1: Principle of NFCV. In the beginning of the procedure, 2 short cine loops (3 sec each) are recorded and are used as the dynamic background for catheter visualization. Specially-designed catheters with miniaturized sensors at the tip are inserted in the patient and visualized by the NFCV system. Please click here to view this video.
Discussion
Radiation exposure for interventional cardiologists and electrophysiologists is an underestimated risk because of its unpredictable side-effects. Current literature reveals a higher incidence of left-sided brain-tumors among this subgroup of clinicians, suggesting that the proximity of the left hemisphere to the X-Ray source may be a culprit12. The latency between radiation exposure and diagnosis of neoplasia has been reported to be 20 years or more. Therefore, today’s interventionalists should use all technological options to reduce radiation exposure to a minimum.
The NFCV system can help reduce fluoroscopy exposure without affecting procedure time14,15 with a workflow that was adapted several times over the past 3 years in order to minimize radiation exposure according to the ALARA principle.
3D mapping systems can help to improve the understanding of complex 3-dimensional structures, but the basic orientation for the operator is generated using conventional fluoroscopy.
The transseptal puncture remains the largest contributing step (75-80%) of the radiation dose during these procedures since no sensor-equipped material for use with NFCV technology is currently available. Especially in unexperienced hands this represents the most critical step in that procedure- other imaging modalities (such as intracardiac or transesophageal echo) can contribute to safe punctures and low complication rates.
The NFCV is not only used in ablation procedures but also in complex implantations such as cardiac resynchronization therapy (CRT). In these procedures, the system allows the reduction of fluoroscopy burden by 75-80% compared to conventional implantations13. A recent publication could show that after a learning curve of 30 – 40 procedures a median fluoroscopy time of 1.1 min for 50 consecutive patients is feasible and safe14. This was confirmed when extending the data acquisition to >500 patients (see Figure 4).
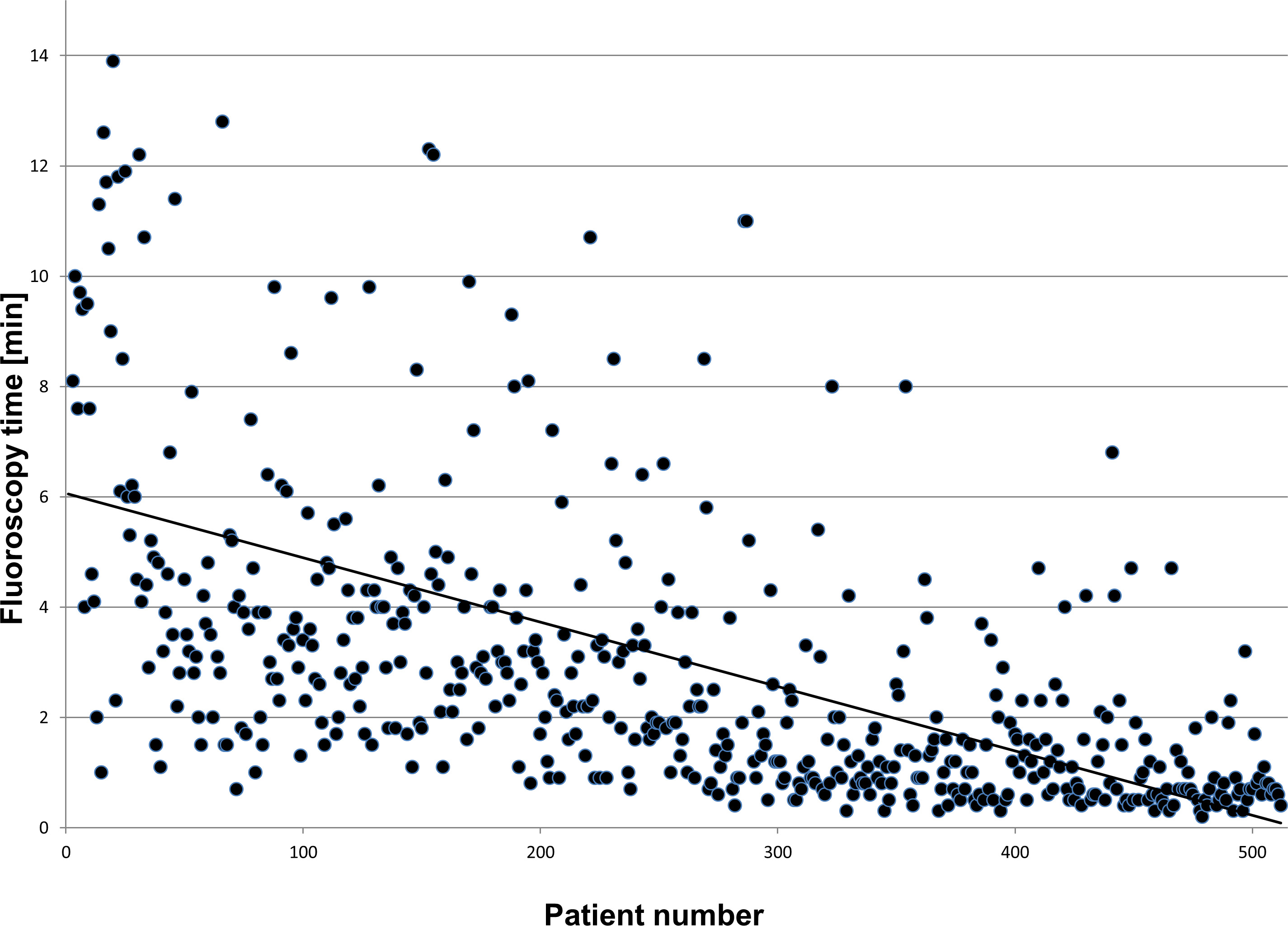
Figure 4: Please click here to view a larger version of this figure.
The limitation of the current available system is that only the tips of the catheters are visualized. Unexperienced operators will probably not be able to interpolate from the orientation of the tip to know what the position of the catheter shaft will be. Furthermore, the system is not able to visualize the transseptal sheath yet. Only a limited choice of catheters are currently available- therefore only a limited number of different procedures is suitable using NFCV technology.
In near future more devices and tools will be available that are equipped with a sensor to be visualized non-fluoroscopically. The system here basically works as a cardiovascular platform for different procedures; electrophysiology is just the first application that has been introduced.
Disclosures
The authors have nothing to disclose.
Acknowledgements
We acknowledge the funding by SJM.
Materials
| MediGuide System | SJM | MG1000 | Non fluoroscopic mapping system |
| Patient Reference Sensor (PRS) Patch | SJM | H700071 | Reference sensor |
| Livewire™ Diagnostic Catheter MediGuide Enabled™ | SJM | D402058 | diagnostic catheter |
| Agilis Nxt steerable introducers 71cm small curle | SJM | 408309 | steerable sheath |
| BRK transseptal needle and stainless steel stylet | SJM | 408314 | transseptal needle |
| EnSite Velocity patch set | SJM | 100003331 | 3D mapping tools |
| Safire BLU | SJM | A088087 | Ablation catheter |
| Sensitherm | SJM | 26155ST | thermoprobe |
| Siemens Artis | Siemens | x | X Ray biplanar |
| Ensite Velocity v. 2.1 | SJM | x | 3D mapping system |
| Ampere generator | SJM | H700494 | RF generator |
| Ampere Remote control | SJM | H700490 | Remote control for generator |
| Cool point | SJM | IBI-89003 | Irrigation pump |
| Cool point tubing set | SJM | 85785 | Tubing set |
References
- Caponi, D., et al. Ablation of atrial fibrillation: Does the addition of three-dimensional magnetic resonance imaging of the left atrium to electroanatomic mapping improve the clinical outcome?), A randomized comparison of carto-merge vs. Carto-xp three-dimensional mapping ablation in patients with paroxysmal and persistent atrial fibrillation. Europace. 12, 1098-1104 (2010).
- Flugelman, M. Y., et al. Medical positioning system: A technical report. EuroIntervention. 4, 158-160 (2008).
- Jeron, A., et al. First-in-man (fim) experience with the magnetic medical positioning system (mps) for intracoronary navigation. EuroIntervention. 5, 552-557 (2009).
- Sommer, P., Rolf, S., Richter, S., Hindricks, G., Piorkowski, C. non-fluoroscopic catheter tracking: The mediguide system. Herzschrittmacherther Elektrophysiol. 23, 289-295 (2012).
- Sommer, P., et al. Initial experience in ablation of typical atrial flutter using a novel three-dimensional catheter tracking system. Europace. 15, 578-581 (2013).
- Sommer, P., et al. Mediguide in supraventricular tachycardia: Initial experience from a multicentre registry. Europace. 15, 1292-1297 (2013).
- Rolf, S., et al. Catheter ablation of atrial fibrillation supported by novel nonfluoroscopic 4d navigation technology. Heart Rhythm. 10, 1293-1300 (2013).
- Piorkowski, C., et al. Steerable versus nonsteerable sheath technology in atrial fibrillation ablation: A prospective, randomized study. Circ Arrhythm Electrophysiol. 4, 157-165 (2011).
- Eitel, C., et al. Circumferential pulmonary vein isolation and linear left atrial ablation as a single-catheter technique to achieve bidirectional conduction block: The pace-and-ablate approach. Heart Rhythm. 7, 157-164 (2010).
- Halm, U., et al. Thermal esophageal lesions after radiofrequency catheter ablation of left atrial arrhythmias. Am J Gastroenterol. 105, 551-556 (2010).
- Vallakati, A., et al. Impact of nonfluoroscopic mediguide tracking system on radiation exposure in radiofrequency ablation procedures (less-rads registry)-an initial experience. J Interv Card Electrophysiol. 38, 95-100 (2013).
- Roguin, A., Goldstein, J., Bar, O., Goldstein, J. A. Brain and neck tumors among physicians performing interventional procedures. Am J Cardiol. 111 (9), 1368-1372 (2013).
- Richter, S., et al. Cardiac resynchronization therapy device implantation using a new sensor-based navigation system: results from the first human use study. Circ Arrhythm Electrophysiol. 6 (5), 917-923 (2013).
- Sommer, P., et al. Non-fluoroscopic catheter visualization in AF ablation: experience from 375 consecutive procedures. Circ Arrhythm Electrophysiol. 7 (5), 869-874 (2014).
- Sommer, P., Richter, S., Hindricks, G., Rolf, S. Non-fluoroscopic catheter visualization using MediGuide technology: experience from the first 600 procedures. J Interv Card Electrophysiol. 40 (3), 209-214 (2014).
- Eitel, C., et al. Circumferential pulmonary vein isolation and linear left atrial ablation as a single-catheter technique to achieve bidirectional conduction block: the pace-and-ablate approach. Heart Rhythm. 7 (2), 157-164 (2010).
- Rolf, S., et al. Tailored Atrial Substrate Modification Based On Low-Voltage Areas in Catheter Ablation of Atrial Fibrillation. Circ Arrythm Electrophysiol. 7, 483-489 (2014).

