Osteoclast Derivation from Mouse Bone Marrow
Summary
Osteoclasts are the principal bone-resorbing cell in the body. An ability to isolate osteoclasts in large numbers has resulted in significant advances in the understanding of osteoclast biology. In this protocol, we describe a method for isolation, cultivating and quantifying osteoclast activity in vitro.
Abstract
Osteoclasts are highly specialized cells that are derived from the monocyte/macrophage lineage of the bone marrow. Their unique ability to resorb both the organic and inorganic matrices of bone means that they play a key role in regulating skeletal remodeling. Together, osteoblasts and osteoclasts are responsible for the dynamic coupling process that involves both bone resorption and bone formation acting together to maintain the normal skeleton during health and disease.
As the principal bone-resorbing cell in the body, changes in osteoclast differentiation or function can result in profound effects in the body. Diseases associated with altered osteoclast function can range in severity from lethal neonatal disease due to failure to form a marrow space for hematopoiesis, to more commonly observed pathologies such as osteoporosis, in which excessive osteoclastic bone resorption predisposes to fracture formation.
An ability to isolate osteoclasts in high numbers in vitro has allowed for significant advances in the understanding of the bone remodeling cycle and has paved the way for the discovery of novel therapeutic strategies that combat these diseases.
Here, we describe a protocol to isolate and cultivate osteoclasts from mouse bone marrow that will yield large numbers of osteoclasts.
Introduction
Bone remodeling is dynamic and involves the coupling of bone formation with bone resorption1. This tightly regulated process is responsible for maintaining the skeleton during normal homeostasis, and in response to injury and disease.
Osteoclasts are unique, multinucleated cells that are capable of resorbing both the organic and inorganic matrices of bone. Osteoclasts are derived from the monocyte/macrophage lineage of the bone marrow2-5. Abnormalities in the function or formation of osteoclasts can result in a variety of clinical pathologies, including common conditions like osteoporosis.
The ability to generate osteoclasts in vitro has allowed for significant advances in our understanding of bone biology6. As a result, new therapeutic agents are emerging to treat osteoclast-related diseases which are responsible for significant morbidities and mortalities7. Homeostatic maintenance of bone mass and strength requires the concerted action of bone-forming osteoblasts and bone-resorbing osteoclasts8,9. Bone homeostasis is altered in a number of diseases, including post-menopausal osteoporosis, in which increased osteoclast activity leads to pathogenic loss of bone mass and density10. With increasing availability of transgenic murine models of human disease, there is more opportunity to decipher the role of the osteoclasts in human bone disease11-13.
Numerous protocols for osteoclast culturing techniques appear in the literature, with many variations described9,12,14. Xing and colleagues describe similar methodology to the protocol described below, in their description of osteoclastogenic assays from murine bone marrow cells. However to release the bone marrow cells following long bone harvest, Xing et al. flush the marrow cavity with α-MEM complete media14. Catalfamo examines the effect of hyperglycemia on osteoclast function and describes a method in which all cells mobilized by bone marrow flushing are cultured for 24 hr, at which point the non-adherent cells are discarded12, a technique also used by Boyle et al.9 These previously published protocols necessitate the practice of flushing the bone marrow, a tedious practice, which also introduces the risk of a needle stick injury and loss of valuable bone marrow, as one must cut both ends of the bone. The protocol, which we describe, implements the use of a mortar and pestle to isolate osteoclasts, which is similar to the method of macrophage isolation described by Weischenfeldt et al.15
Our experience, however, is that osteoclast isolation and in vitro culture using previously published techniques results in variable outcomes in terms of osteoclast production, often resulting in an inability to cultivate osteoclasts. Therefore, we have devised a protocol that allows for the consistent isolation of mouse bone marrow to produce large numbers of multinucleated osteoclasts in vitro, with an approximate yield of 70-80% of cells initially plated forming macrophages and subsequently osteoclasts, in the presence of osteoclast induction media.
Protocol
NOTE: Ethical statement: All research involving vertebrate animals was performed in accordance protocols approved by the Stanford Administrative Panel on Laboratory Animal Care (APLAC).
1. Preparation
- Allow 10 ml of commercially available density gradient cell separation media (which contains polysucrose and sodium diatrizoate, adjusted to a density of 1.077 g/ml) to come to RT in a 50 ml conical tube.
- Prepare flow cytometry (FACS) buffer with 1x phosphate buffered saline (PBS) and 2% fetal calf serum (FCS). Take a 50 ml aliquot and maintain this aliquot at RT for use during the density gradient cell separation media step.
- Have a bucket of ice ready to maintain isolated tissue and cells in during the isolation procedure.
2. Prepare Culture Media
- Prepare basal media: MEM (Minimal Essential Medium), without phenol red (500 ml), glutamate (1x), FCS (10x), 10,000 units/ml penicillin (1x).
- Filter basal media with a 0.22 μm filter.
- Prepare bone marrow macrophage induction media:
- Take 50 ml of the basal media prepared in Step 2.1.
- Add Prostaglandin E2 (PGE2, Mr 352.465 g/mol) at 10-7 M final concentration.
- Add Macrophage-Colony Stimulating Factor (M-CSF) at 10 ng/ml.
- Do not filter the final solution of bone marrow macrophage stimulating media.
- Prepare osteoclast induction media:
- Take 50 ml of the basal media prepared in Step 2.1.
- Add Prostaglandin E2 (PGE2, Mr 352.465 g/mol) at 10-7 M final concentration
- Add Macrophage-Colony Stimulating Factor (M-CSF) at 10 ng/ml.
- Add RANKL at 10 ng/ml.
- Do not filter the final solution of osteoclast induction media.
3. Bone Marrow Isolation
- Euthanize one C57BL/6 mouse using carbon dioxide inhalation method, followed by cervical dislocation.
NOTE: Rodents must be euthanized by trained personnel using appropriate technique, equipment and agents. - Position and secure the mouse on a dissection board and spray with 70% alcohol.
- Dissect out the femurs, tibiae, humeri and spine.
- Begin with the mouse in supine position. Make an incision vertically along the lower limb and begin by dissecting along the length of the femur and tibia by dividing soft tissue attachments. Finally, dislocate bones from the knee and hip joint to free bones fully from the skeleton.
- Proceed to make an incision along the length of the upper limb to isolate the humerus. Dislocate the humerus at the shoulder and elbow joints by dissecting soft tissue capsular attachment.
- Finally, turn the mouse over, so that the mouse lies prone. Make a skin incision along the length of the spine, in the midline. Observe the paravertebral soft tissue on each side of the spine and incise along the bony edge of the spine, dissecting through the paravertebral soft tissue mass. Using a scissors, make a horizontal cut across the spine at the base of the neck, and at the end of the spine, just proximal to the tail insertion, freeing the spine from soft tissue attachment.
- Once the bones are harvested, keep them in FACS buffer on ice.
- Use tissue paper to clean muscle and soft tissue from the bones.
- Place the cleaned bones into a pestle and mortar with 3 ml of FACS buffer.
- Crush the bones gently in the pestle and mortar.
- Aspirate off the blood-stained fluid and transfer into a 50 ml conical tube by passing the solution through a 70 μm cell strainer.
- Add 5 ml FACS buffer to the crushed bone and crush further. Again, remove the fluid using a pipette and transfer to the 50 ml conical tube through a 70 μm strainer.
- Typically crush the bones twice to three times in fresh FACS buffer, using a mortar and pestle, each time adding a further 5 ml of fresh FACS buffer, until the fluid does not stain red.
- Centrifuge the bone marrow solution at 200 x g for 5 min at 4 °C to yield a cell pellet.
4. Gradient Separation of Bone Marrow Cells
- Aspirate the supernatant and resuspend in 10 ml of FACS buffer (maintained at RT).
- Take the 50 ml conical tube containing 10 ml of commercially available density gradient cell separation media (RT).
- Layer the bone marrow solution onto the density gradient cell separation media. Do this slowly using an electric pipette. Angle the pipette tip against the inner surface near the top of the conical tube and tilt the conical tube to about 30°. Using a slow speed setting on the pipette, gently expel the bone marrow solution so as not to disturb the density gradient cell separation media.
NOTE: Ultimately there will be a clear definition between the two solutions (cellular solution and density gradient cell separation media solution). - Using a RT centrifuge, centrifuge the density gradient cell separation media and cell solution in a balanced centrifuge at 200 x g for 15 min. In addition, remove acceleration and deceleration on the centrifuge to ensure adequate phase separation using the density gradient cell separation media gradient.
- After centrifugation, aspirate off the cloudy middle layer which contains the bone marrow cells of interest.
- Transfer these cells into a new conical tube. Add 20 ml of additional FACS buffer (kept on ice) to wash the cells.
- Centrifuge at 200 x g for 5 min at 4 °C to yield a cell pellet.
- Resuspend the final cell pellet in 1ml of macrophage stimulating media to allow for cell counting using a hemocytometer.
5. Cell Counting
- Take 10 µl of the cell solution and mix with 10 μl of trypan blue. Load 10 µl of the resultant mix onto a hemocytometer and obtain the cell count per ml of solution.
6. Cell Culturing
- Place 2 ml of macrophage stimulating media into each well of a 24-well plate.
- Add 200,000 cells to each well.
NOTE: In our experience, cell-plating density is one of the most crucial requirements for adequate in vitro culturing of osteoclasts. - Gently agitate the plate and place in a regular incubator at 37 °C.
- Do not change the media for 3 days.
- After 3 days in macrophage stimulating media, change the media to osteoclast media.
- Hereafter, change the media daily for 5–7 days, at which point, large, multinucleated osteoclasts should be visible on the culture plate.
7. Staining with Tartrate Resistant Acid Phosphatase (TRAP)
- After 5–7 days of in vitro culture, aspirate the media from the culture well.
- Wash the wells gently with 1x PBS three times.
- Fix the cells by adding 4% paraformaldehyde solution and leave for 1 hr at 4 °C.
- During this time, prepare the TRAP stain (which is commercially available) by pre-warming to 37 °C.
- After 1 hr in paraformaldehyde, aspirate off paraformaldehyde and wash gently 3 times with 1x PBS.
- Add 1 ml of pre-warmed TRAP stain into each well of the plate to be stained.
- Place the plate in an incubator at 37 °C for 1 hr, shielded from light.
- After 1 hr, aspirate off the TRAP stain.
- Wash the wells 3 times with pre-warmed deionized water.
- Counterstain the well with Gill’s Hematoxylin (CAUTION) for 1–2 min.
- Image osteoclasts using bright field microscopy.
- Wash wells with deionized water until an adequate color intensity of the stain is achieved, typically when nuclei appear blue.
8. Osteoclast Resorption Assay
- Use a commercially available 24-well polystyrene culture dish that is pre-coated with either bone substrate or an inorganic crystalline calcium phosphate coating.
- Place 2 ml of macrophage stimulating media into each well.
- Add 200,000 freshly isolated bone marrow cells obtained at the end of step 2 to each well.
- Gently agitate the plate prior to placing it in an incubator at 37 °C for 3 days.
- On day 3, change the media to osteoclast differentiation media.
- Perform daily media changes with osteoclast differentiation media for 5–7 days.
9. Quantification of Osteoclast Resorption Activity
- After 5–7 days of in vitro culture in osteoclast differentiation media on a mineralized substrate, analyze the mineralized surface, as described below, to assess the ability of osteoclasts to form resorption pits, an indicator of osteoclastic activity.
- Aspirate media from the resorption assay plate and add 100 μl of 10% bleach solution to each well.
- Incubate the cells with the 10% bleach solution for 15 min at RT.
- Wash the wells 3 times with distilled water.
- Aspirate off any remaining water and shake off excess water from the plate and allow to air dry for 3–5 hr.
- Counterstain the plate with a commercially available Von Kossa staining kit to allow for easier visualization of the un-resorbed substrate.
- Use bright field microscopy to take images of representative parts of the resorbed surface.
- Using image analysis software, calculate the percentage of the resorbed surface to allow for quantification of osteoclastic resorptive activity.
Representative Results
The aim of this method was to easily isolate large numbers of osteoclasts in vitro, typically in one week. Successful isolation of large numbers of osteoclasts was confirmed using tartrate-resistant acid phosphatase staining (Figure 1A). Large osteoclasts are visualized as large purple cells with multiple nuclei (typically ≥ 3 nuclei). Using this protocol, it is common to isolate osteoclasts with as many as 30 nuclei per osteoclast (Figure 1B).
Using a mineralized surface is considered the gold standard method of assessing osteoclast resorptive activity in vitro. Using this method, the percentage of mineralized surface, or “resorption pits” that have formed allows for determination of osteoclast resorptive capacity (Figure 2).
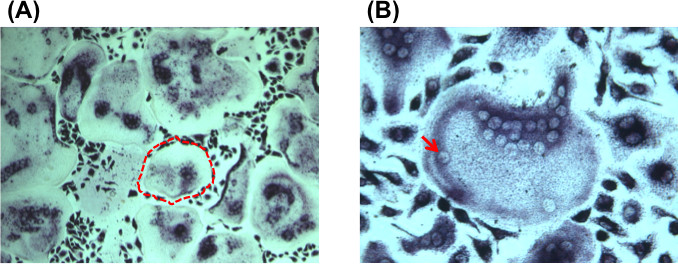
Figure 1. (A) Tartrate resistant acid phosphatase staining of cultured bone marrow following 10 days in culture under osteoclast inductive conditions. Bright field micrograph at 10X magnification demonstrates multiple large, multinucleated osteoclasts that are TRAP positive (purple staining). An example of a large, multinucleated osteoclast is outlined by a red dashed line. (B) Bright field micrograph at 10X magnification demonstrating a large, multinucleated, TRAP positive osteoclast. An example of a nucleus within a multinucleated osteoclast is shown by the red arrow. Please click here to view a larger version of this figure.

Figure 2. Bright field micrograph at 10X magnification demonstrates resorption pits formed by osteoclastic resorption activity. Mineralized matrix is stained with Von Kossa stain to allow for visualization. The black dashed lines outline an example of resorbed areas of matrix. Please click here to view a larger version of this figure.
Discussion
An ability to easily isolate and cultivate large numbers of osteoclasts in vitro has been responsible for helping to advance understanding of bone biology and osteoclast-mediated diseases. It was the identification of RANKL that lead to this, when it was recently identified as the major regulator of osteoclast formation, differentiation and survival16-18.
It has been our experience that the in vitro cultivation of osteoclasts from bone marrow is largely dependent on seeding density. We observed that failure of this protocol to yield osteoclasts is typically due to a suboptimal seeding density when plating bone marrow-derived cells. Thus, in this protocol, it is recommended to seed 200,000 cells per 24-well plate well. Furthermore, in order to achieve maximum success with this technique, it is recommended to prepare the osteoclast induction media precisely as described above and to change the media at the same time each day, after the initial 3 days when one does not need to disturb the culture plates. This may allow a constant concentration of the osteoclast induction media factors. In addition, we have found that the use of specific reagents also allows greater potential for culturing of large numbers of multinucleated osteoclasts.
This technique is limited by the availability of a RT centrifuge, in which one can alter acceleration and deceleration parameters and procurement of specific reagents. It is otherwise a very straightforward protocol, with which we have had much success, in a range of mice of differing age and genetic background (e.g., C57BL/6, CD1, NOD SCID, Lep db-/-).
This technique offers a reliable method to generate osteoclasts in vitro and to determine their ability to resorb mineralized matrix. Using this protocol, it is possible to isolate large numbers of multinucleated osteoclasts in vitro typically in one week. Our experience is that osteoclast isolation and in vitro culture using previously published techniques results in highly variable outcomes in terms of osteoclast production, often resulting in an inability to cultivate osteoclasts9,12,14.
Osteoclast function is complex and bone resorption is a highly orchestrated process that is mediated at the local level by cross-talk between osteoblasts and osteoclasts19. In addition to local control of bone balance, emerging evidence suggests that osteoclast activity is governed systemically by factors related to immune, neuronal, and other systematic factors20-24.
As the understanding of osteoclast biology continues to advance rapidly, including their diverse role in processes ranging from bone remodeling to their role in regulating cancer metastases to bone25-27, this protocol will allow researchers to consistently isolate large quantities of osteoclasts in vitro.
Disclosures
The authors have nothing to disclose.
Acknowledgements
We acknowledge the support of NIH grants R01 DE021683, R01 DE019434, U01 HL099776, The Oak Foundation and The Hagey Laboratory for Pediatric Regenerative Medicine.
Materials
| Name of the Material/Equipment | Company | Catalog Number | Comments/Description |
| MEM, no glutamine, no phenol red | Gibco | 51200-038 | |
| M-CSF, recombinant mouse | Gibco | PMC2044 | |
| Recombinant Mouse TRANCE/RANK L/TNFSF11 (E. coli expressed) | R&D Systems | 462-TEC-010 | |
| Prostaglandin E2 | Sigma-Aldrich | ||
| Histopaque-1077 | Sigma-Aldrich | 10771 | |
| Acid Phosphatase, Lekocyte (TRAP) kit | Sigma-Aldrich | 387A | |
| Osteoassay bone resorption plates, 24 well plates | Corning Life Sciences | 3987 |
References
- Sims, N. A., Martin, T. J. Coupling the activities of bone formation and resorption: a multitude of signals within the basic multicellular unit. BoneKEy reports. 3, 481 (2014).
- Kahn, A. J., Simmons, D. J. Investigation of cell lineage in bone using a chimaera of chick and quial embryonic tissue. Nature. 258, 325-327 (1975).
- Walker, D. G. Bone resorption restored in osteopetrotic mice by transplants of normal bone marrow and spleen cells. Science. 190, 784-785 (1975).
- Burger, E. H., et al. In vitro formation of osteoclasts from long-term cultures of bone marrow mononuclear phagocytes. The Journal of experimental medicine. 156, 1604-1614 (1982).
- Underwood, J. C. From where comes the osteoclast. The Journal of pathology. 144, 225-226 (1984).
- Lacey, D. L., et al. Bench to bedside: elucidation of the OPG-RANK-RANKL pathway and the development of denosumab. Nature reviews. Drug discovery. 11, 401-419 (2012).
- Brown, J. E., Coleman, R. E. Denosumab in patients with cancer-a surgical strike against the osteoclast. Nature reviews. Clinical oncology. 9, 110-118 (2012).
- Khosla, S. Minireview: the OPG/RANKL/RANK system. Endocrinology. 142, 5050-5055 (2001).
- Boyle, D. L., et al. Differential roles of MAPK kinases MKK3 and MKK6 in osteoclastogenesis and bone loss. PloS one. 9, (2014).
- Hofbauer, L. C., Heufelder, A. E. Role of receptor activator of nuclear factor-kappaB ligand and osteoprotegerin in bone cell biology. Journal of molecular medicine (Berlin, Germany). 79, 243-253 (2001).
- Teramachi, J., et al. Increased IL-6 Expression in Osteoclasts is Necessary but not Sufficient for the Development of Paget’s Disease of Bone. Journal of bone and mineral research : the official journal of the American Society for Bone and Mineral Research. , (2013).
- Catalfamo, D. L., et al. Hyperglycemia induced and intrinsic alterations in type 2 diabetes-derived osteoclast function. Oral diseases. 19, 303-312 (2013).
- Schueler, J., et al. Intratibial injection of human multiple myeloma cells in NOD/SCID IL-2Rgamma(null) mice mimics human myeloma and serves as a valuable tool for the development of anticancer strategies. PloS one. 8, (2013).
- Xing, L., Boyce, B. F. RANKL-Based Osteoclastogenic Assays from Murine Bone Marrow Cells. Methods in molecular biology (Clifton, N.J). 1130, 307-313 (2014).
- Weischenfeldt, J., Porse, B. Bone Marrow-Derived Macrophages (BMM): Isolation and Applications. CSH protocols. , (2008).
- Yamamoto, Y., et al. Osteoblasts provide a suitable microenvironment for the action of receptor activator of nuclear factor-kappaB ligand. Endocrinology. 147, 3366-3374 (2006).
- Yasuda, H., et al. Osteoclast differentiation factor is a ligand for osteoprotegerin/osteoclastogenesis-inhibitory factor and is identical to TRANCE/RANKL. Proceedings of the National Academy of Sciences of the United States of America. 95, 3597-3602 (1998).
- Lacey, D. L., et al. Osteoprotegerin ligand is a cytokine that regulates osteoclast differentiation and activation. Cell. 93, 165-176 (1998).
- Teitelbaum, S. L., Ross, F. P. Genetic regulation of osteoclast development and function. Nature reviews. Genetics. 4, 638-649 (2003).
- Agas, D., Sabbieti, M. G., Marchetti, L. Endocrine disruptors and bone metabolism. Archives of toxicology. 87, 735-751 (2013).
- Manolagas, S. C., O’Brien, C. A., Almeida, M. The role of estrogen and androgen receptors in bone health and disease. Nature Reviews Endocrinology. 9, 699-712 (2013).
- Martin, T. J., Udagawa, N. Hormonal regulation of osteoclast function. Trends in endocrinology and metabolism. 9, 6-12 (1998).
- Nakamura, T., et al. Estrogen prevents bone loss via estrogen receptor alpha and induction of Fas ligand in osteoclasts. Cell. 130, 811-823 (2007).
- Bellido, T., et al. Regulation of interleukin-6, osteoclastogenesis, and bone mass by androgens. The role of the androgen receptor. The Journal of clinical investigation. 95, 2886-2895 (1995).
- Roato, I. Interaction among cells of bone, immune system, and solid tumors leads to bone metastases. Clinica., & developmental immunology. 2013, (2013).
- Autio, K. A., Morris, M. J. Targeting bone physiology for the treatment of metastatic prostate cancer. Clinical advances in hematolog., & oncology. 11, 134-143 (2013).
- Sottnik, J. L., Keller, E. T. Understanding and targeting osteoclastic activity in prostate cancer bone metastases. Current molecular medicine. 13, 626-639 (2013).

