A subscription to JoVE is required to view this content. Sign in or start your free trial.
JoVE Journal
Biology
Hydrodynamic Renal Pelvis Injection for Non-viral Expression of Proteins in the Kidney
Chapters
- 00:05Title
- 00:52Preparation for Surgery
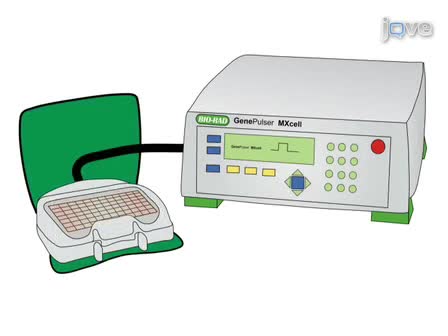
- 01:39Injection Surgery
- 04:03Injection Efficiency and Transgene Effects Assessment
- 06:06Results: Hydrodynamic Renal Pelvis Injection Results in Kidney-specific Gene Expression
- 07:25Conclusion
This protocol describes a method to inject plasmid DNA into the mouse kidney via the renal pelvis to produce transgene expression specifically in the kidney.