14.7: 未知数小x假设
如果反应的平衡常数较小,则平衡位置有利于反应物。 在这种反应中,如果反应物的初始浓度高而 K c 值小,则浓度的变化可忽略不计。 在这种情况下,平衡浓度大约等于其初始浓度。 通过假设某些平衡浓度等于初始浓度,可以使用此估计值简化平衡计算。 但是,要做出此假设,弱酸或弱碱(即 x )的浓度变化必须小于其初始浓度的5%。 如果 x 大于5%,则需要使用二次方程式来求解平衡方程。  
使用代数简化假设计算平衡浓度 平衡中 0.15 M 溶液的 HCN 浓度是多少? 使用 x 来表示平衡上每个生成物的浓度,就可以获得此 ICE表格。 将平衡浓度项代入Kc表达式 重新排列为二次式并为 x 求解 Thus, [H+] = [CN–] = x = 8.6 × 10–6 M and [HCN] = 0.15 – x = 0.15 M. 在这种情况下,浓度变化明显小于初始浓度( K 较小的结果),因此初始浓度变化可忽略不计:
此近似值允许使用更方便的数学方法进行计算,而无需求解二次方程的根:
x 的计算值确实比初始浓度小
,因此近似值是合理的。 如果此简化方法要产生 x 的值而不能证明近似值合理,则需要在不进行近似值的情况下重复进行计算。
本文改编自 Openstax, 化学 2e, 第13.4节:平衡计算。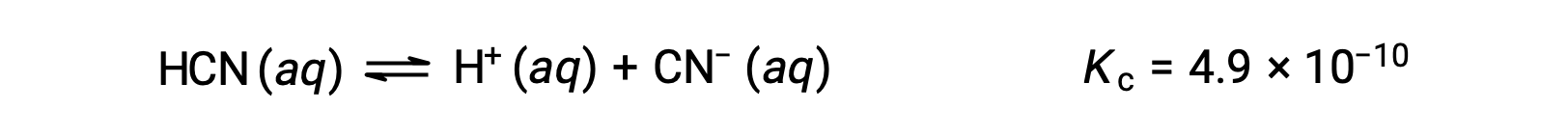
HCN (aq)
H+ (aq)
CN− (aq)
初始浓度 (M)
0.15
0
0
变化 (M)
−x
+x
+x
平衡浓度 (M)
0.15 − x
x
x
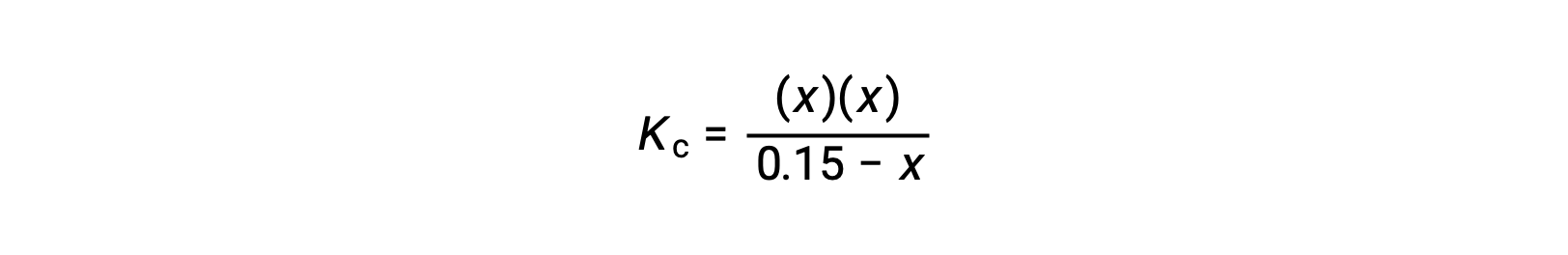
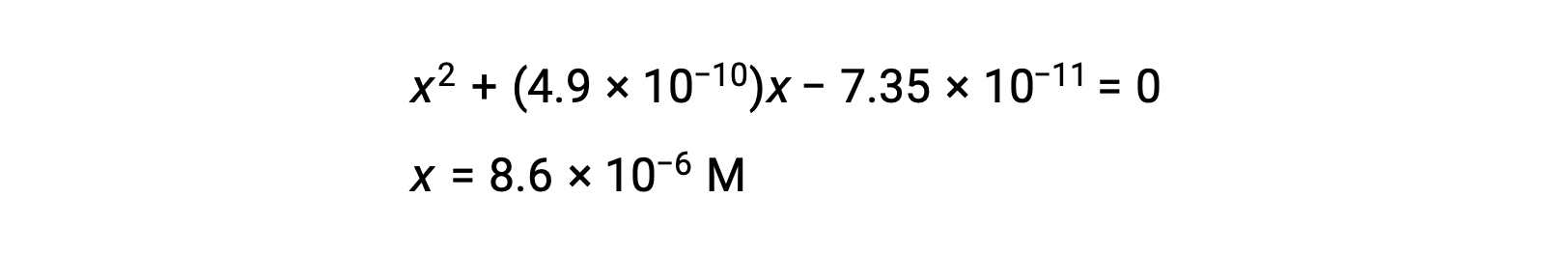

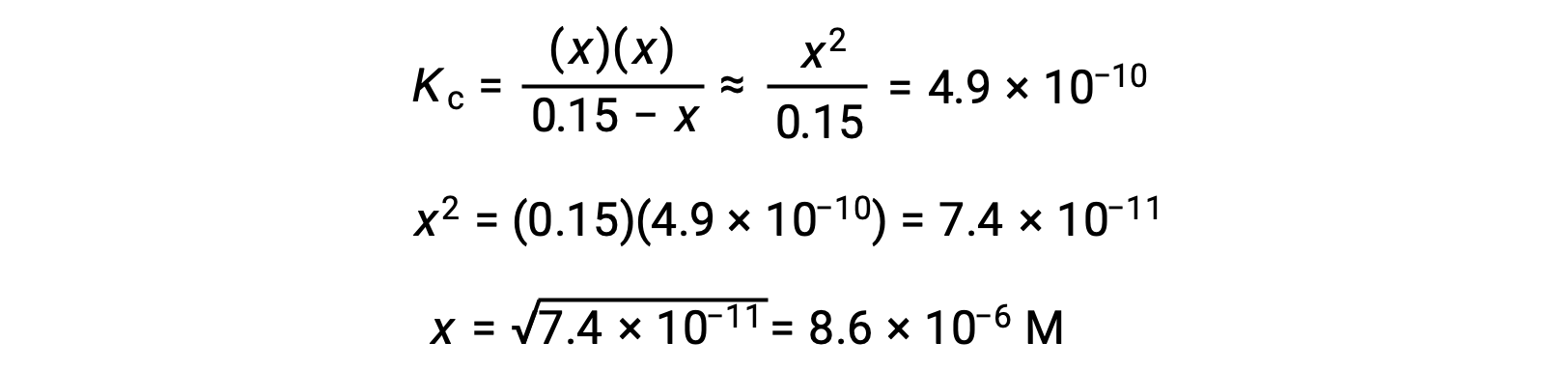
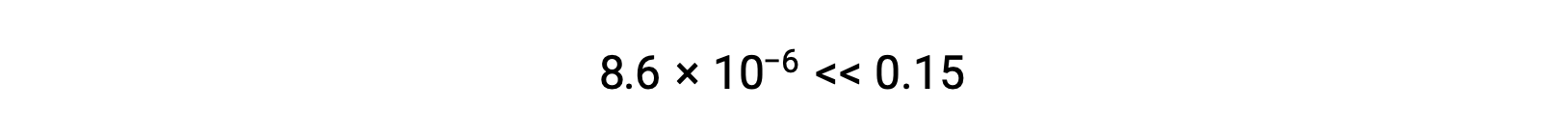
Tags
Small X Assumption
Equilibrium Expression
Quadratic Formula
Equilibrium Constant
Initial Concentrations
Reactants
Products
Approximation
Change In Concentration
ICE Table
Carbonyl Dichloride
Carbon Monoxide
Chlorine Gas


